Buy 4-HO-NiPT (4-hydroxy-N-isopropyltryptamine) Online
Buy 4-HO-NiPT (4-hydroxy-N-isopropyltryptamine) Online
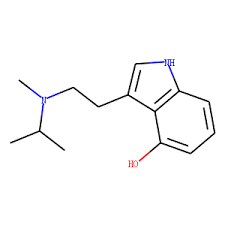
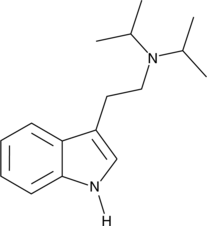
4-HO-NiPT, also known as 4-hydroxy-N-isopropyltryptamine, is a serotonin receptor modulator and putative psychedelic drug of the tryptamine and 4-hydroxytryptamine families related to psilocin (4-HO-DMT).[1][2][3] It is an analogue of 4-HO-MiPT (miprocin) and 4-HO-DiPT (iprocin) and a derivative of norpsilocin (4-HO-NMT) and 4-HO-NET.[2] The drug has been encountered online as a possible novel designer drug.[4]
Use and effects
4-HO-NiPT was not included nor mentioned in Alexander Shulgin‘s book TiHKAL (Tryptamines I Have Known and Loved).[5]
Interactions
Pharmacology
Pharmacodynamics
4-HO-NiPT shows affinity for serotonin receptors, including for the serotonin 5-HT1D, 5-HT1E, 5-HT2A, 5-HT2B, 5-HT2C, 5-HT6, and 5-HT7 receptors (Ki = 229–2,461 nM).[2] Conversely, it did not show affinity for the serotonin 5-HT1B or 5-HT5A receptors, whereas the serotonin 5-HT1A receptor was not reported.[2] 4-HO-NiPT is a partial agonist of the serotonin 5-HT2A, 5-HT2B, and 5-HT2C receptors, with EC50Tooltip half-maximal effective concentration (EmaxTooltip maximal efficacy) values of 24 nM (88.4%), 188 nM (69.9%), and 963 nM (45.2%), respectively.[2] It was also a weak agonist of other serotonin receptors, including the serotonin 5-HT1B, 5-HT1E, 5-HT1F, and 5-HT7A receptors (EC50 = 1,400–23,000 nM, Emax = 20.4–123%), but not of the 5-HT1A, 5-HT1D, 5-HT4, 5-HT5A, or 5-HT6 receptors.[2]
In contrast to norpsilocin, but similarly to psilocin and certain other N-monoalkyltryptamines like 4-HO-NET, 4-HO-NPT, 4-HO-NALT, and 4-HO-NBnT, the drug produces the head-twitch response, a behavioral proxy of psychedelic effects, in rodents.[2] It showed 30-fold lower potency than psilocin in this action in mice, but produced about the same maximal response.[2] Similarly to 4-HO-NiPT, other N-monoalkyltryptamines were also much less potent than psilocin, in the range of 10- to 26-fold less potent.[2] In addition to the head-twitch response, 4-HO-NiPT produces hypothermia in rodents.[2]
4-HO-NiPT is a known metabolite of 4-AcO-DiPT and presumably also of 4-HO-DiPT.[1]
Chemistry
Synthesis
The chemical synthesis of 4-HO-NiPT has been described.[1][2]
Analogues

Analogues of 4-HO-NiPT include 4-hydroxytryptamine (4-HT or 4-HO-T), 4-HO-MiPT (miprocin), 4-HO-DiPT (diprocin), psilocin (4-HO-DMT), norpsilocin (4-HO-NMT), 4-HO-NET, 4-HO-NPT, 4-HO-NALT, and 4-HO-NBnT, among others.[2]
History
4-HO-NiPT was first described in the scientific literature, as a metabolite of 4-AcO-DiPT, in 2022.[6] Subsequently, its synthesis was described in 2023[1] and its pharmacology was reported in 2024.[2] The drug was reported as a possible novel designer drug online in 2022.[4]
Reviews
There are no reviews yet.