Buy CYB003 (CYB-003) Online
Buy CYB003 (CYB-003) Online
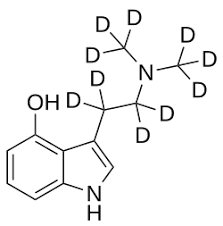
CYB003, or CYB-003, also known as deuterated psilocybin analogue, is a serotonergic psychedelic related to psilocybin which is under development for the treatment of major depressive disorder, alcoholism, and other psychiatric disorders.[1][4][5][6][7][2][3] It is taken orally.[1] The drug is a tryptamine derivative and is a deuterated analogue of psilocybin and psilocin.[1][5][6][2]
Interactions
Pharmacology
The pharmacodynamic profile of CYB003, including its interactions with serotonin receptors and its effects in animals, is similar to that of psilocin.[2] As with psilocin, CYB003 is a potent agonist of the serotonin 5-HT2A receptor and produces psychedelic-like effects in animals.[2] However, it was developed to have improved pharmacokinetic properties compared to psilocybin, including reduced variability in circulating levels, a faster onset of action, and a shorter duration.[3]
Chemistry

The exact chemical structure of CYB003 (i.e., which specific hydrogen atoms have been deuterated) does not yet seem to have been disclosed.[6][5] However, an INNTooltip International Nonproprietary Name of deupsilocin for a deuterated form of psilocin called d10-psilocin (decadeuteropsilocin) with CAS number 1435934-64-7 was proposed in 2023 and recommended in 2024.[8][9][10][11] This deuterated analogue of psilocin has been patented by Cybin as well as by Lennham Pharmaceuticals.[12][13][10] Cybin has also patented other deuterated psilocin analogues.[14] Other deuterated drugs related to CYB003 or deupsilocin/d10-psilocin include the deuterated DMT analogues CYB004 and SPL028 and the deuterated phenethylamine CYB005.[5][15]
Research
In 2024, CYB003 received a breakthrough therapy designation from the U.S. FDA[16] and was in phase 3 clinical trials for major depressive disorder and is in the preclinical stage of development for alcoholism and other psychiatric disorders.[1][4][17] Two phase 3 clinical trials for major depressive disorder are being initiated in November 2024 and February 2025.[1][4][17] The drug is under development by Cybin.[1][4][17]
Reviews
There are no reviews yet.