Buy 5-Methoxy-2 Cas 67292-68-6
Buy 5-Methoxy-2 Cas 67292-68-6
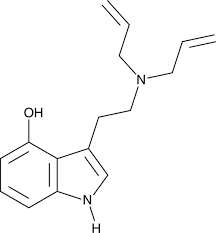
5-Methoxy-2,N,N-trimethyltryptamine (5-MeO-2,N,N-TMT, 5-MeO-TMT), also known as 2-methyl-5-methoxy-N,N-dimethyltryptamine (2-Me-5-MeO-DMT), is a serotonin receptor modulator and psychedelic drug of the tryptamine and 2-alkyltryptamine families.[1][3][4][2][5] It is taken orally.[1][2]
5-MeO-2-TMT was first synthesized by Alexander Shulgin and reported in his 1997 book TiHKAL (Tryptamines I Have Known and Loved).[1]
Use and effects
According to Alexander Shulgin in his book TiHKAL (Tryptamines I Have Known and Loved), 5-MeO-TMT has a dose range of 75 to 150 mg orally and a duration of 5 to 10 hours.[1] It produces effects including sexual stimulation, enhanced orgasm, relaxation, sedation, tingling, sleep disturbances, chills and cold sensations, time dilation, reduced heart rate, reduced respiratory rate, mild nausea, motor incoordination, visual waviness, mild to pronounced closed-eye visuals, emotional lability, crying, body temperature fluctuations, uncomfortableness, gastrointestinal disturbances, and abdominal pain.[1] It has been said that 5-MeO-TMT at a dose of 150 mg is definitely hallucinogenic and can be compared to a moderate 300 mg dose of mescaline.[1]
Interactions
Pharmacology
Pharmacodynamics
| Target | Ki (nM) |
|---|---|
| 5-HT1A | 200 |
| 5-HT1B | >10,000 |
| 5-HT1D | 250 |
| 5-HT1E | 1,800 |
| 5-HT1F | ND |
| 5-HT2A | >10,000 (rat) |
| 5-HT2B | ND |
| 5-HT2C | 4,020 (rat) |
| 5-HT3 | ND |
| 5-HT4 | ND |
| 5-HT5A | 10,450 |
| 5-HT6 | 60–80 |
| 5-HT7 | 145 |
| α1A–α2C | ND |
| β1–β3 | ND |
| D1–D5 | >10,000 |
| H1 | >10,000 |
| H2 | ND |
| H3, H4 | >10,000 |
| M1–M5 | >10,000 |
| I1 | ND |
| σ1, σ2 | ND |
| TAAR1Tooltip Trace amine-associated receptor 1 | ND |
| SERTTooltip Serotonin transporter | >10,000 |
| NETTooltip Norepinephrine transporter | 6,380 |
| DATTooltip Dopamine transporter | >10,000 |
| Notes: The smaller the value, the more avidly the drug binds to the site. All proteins are human unless otherwise specified. Refs: [6][3][5] | |
The affinities of 5-MeO-TMT for numerous targets have been reported.[3][5] 2-Methyltryptamines like 5-MeO-TMT show a loss of affinity for the serotonin 5-HT2A receptor but retained affinity for the serotonin 5-HT6 receptor.[4][5][3] It also retains significant affinity for the serotonin 5-HT1A, 5-HT1D, and 5-HT7 receptors.[3][5] In contrast to 5-MeO-DMT, 5-MeO-TMT is orally active, suggesting that the 2-methyl group blocks metabolism by monoamine oxidase (MAO).[1]
Chemistry
Synthesis
The chemical synthesis of 5-MeO-2-TMT has been described.[1]
Analogues
- 2-Methyltryptamine (2-MT or 2-Me-T)
- 2-Methyl-N,N-diethyltryptamine (2-Me-DET)
- 2,N,N-Trimethyltryptamine (2,N,N-TMT or 2-Me-DMT)
- 2,α-Dimethyltryptamine (2,α-DMT or 2-Me-αMT)
- 5-Methoxy-7,N,N-trimethyltryptamine (5-MeO-7-TMT or 7-Me-5-MeO-DMT)
- 2-Ethyl-5-methoxy-N,N-dimethyltryptamine (EMDT; 2-Et-5-MeO-DMT)
- BGC20-761 (5-MeO-2-phenyl-DMT)
Society and culture
Legal status
Canada
5-MeO-2-TMT is not an explicitly nor implicitly controlled substance in Canada as of 2025.[7]
United States
5-MeO-2-TMT is not an explicitly controlled substance in the United States.[8] However, it could be considered a controlled substance under the Federal Analogue Act if intended for human consumption.

Reviews
There are no reviews yet.