Buy 5-MeO-DALT Cas 928822-98-4
Buy 5-MeO-DALT Cas 928822-98-4
5-MeO-DALT, also known as N,N-diallyl-5-methoxytryptamine or as foxtrot, is a psychedelic drug of the tryptamine and 5-methoxytryptamine families.[1][3] It is taken orally.[1]
5-MeO-DALT was first synthesized and described by Alexander Shulgin, who disclosed the compound in 2004.[1][4] It has been encountered as a novel designer and recreational drug.[4][5][6]
Use and effects
According to Alexander Shulgin in a follow-up entry to his book TiHKAL (Tryptamines I Have Known and Loved), the dose of 5-MeO-DALT is 12 to 20 mg orally and its duration is 2 to 4 hours.[1][7] A wider dose range of 12 to 25 mg has also been reported.[8] It is said to onset and peak remarkably quickly via the oral route, with an onset of less than 15 minutes and a time to peak of 30 minutes.[1] The effects of 5-MeO-DALT were reported by Shulgin to include positive emotional changes, lightheadedness, increased appreciation of music and sex, and closed-eye visuals.[1] There was said to be a lack of open-eye visuals and it was said to be relatively light in psychedelic character.[1]
Overdose
There is little published literature on the toxicity of 5-MeO-DALT.[9] Case reports of overdose have been published, with effects including loss of consciousness, visual hallucinations, acute delirium, and rhabdomyolysis, among others.[9][10][11] A death related to behavioral intoxication has been reported.[3]
Interactions
Pharmacology
Pharmacodynamics
| Target | Affinity (Ki, nM) |
|---|---|
| 5-HT1A | 3.26–48 (Ki) 2.9–3.4 (EC50Tooltip half-maximal effective concentration) 99–102% (EmaxTooltip maximal efficacy) |
| 5-HT1B | 551–735 |
| 5-HT1D | 53–107 |
| 5-HT1E | 322–500 |
| 5-HT1F | ND |
| 5-HT2A | 48–218 (Ki) 8.4–139.4 (EC50) 91–114% (Emax) |
| 5-HT2B | 45–59 (Ki) 18–33 (EC50) 86–90% (Emax) |
| 5-HT2C | 456–1,083 (Ki) 75–299a (EC50) 88–99%a (Emax) |
| 5-HT3 | >10,000 |
| 5-HT4 | ND |
| 5-HT5A | 3,312 |
| 5-HT6 | 87–153 |
| 5-HT7 | 87–90 |
| α1A–α1D | >10,000 |
| α2A | 215–228 |
| α2B | 726–956 |
| α2C | 1,467–641 |
| β1–β3 | >10,000 |
| D1–D2 | >10,000 |
| D3 | 699 |
| D4–D5 | >10,000 |
| H1 | 505–1,373 |
| H2 | 4,250–>10,000 |
| H3 | 2,820 (human) 1,712 (guinea pig) |
| H4 | >10,000 |
| M1–M5 | >10,000 |
| nAChTooltip Nicotinic acetylcholine receptor | >10,000 |
| I1 | ND |
| σ1 | 333 (human) 301–398 (rodent) |
| σ2 | 340 (human) 253 (rat) |
| TAAR1Tooltip Trace amine-associated receptor 1 | ND |
| MORTooltip μ-Opioid receptor, DORTooltip δ-Opioid receptor | >10,000 |
| KORTooltip κ-Opioid receptor | 899–1,132 |
| SERTTooltip Serotonin transporter | 499–1,408 (Ki) >100,000 (IC50Tooltip half-maximal inhibitory concentration) (rat) 930–22,313 (IC50) (human) >100,000 (EC50) (rat) |
| NETTooltip Norepinephrine transporter | >10,000 (Ki) >100,000 (IC50) (rat) >100,000 (EC50) (rat) |
| DATTooltip Dopamine transporter | 3,378 (Ki) >100,000 (IC50) (rat) >100,000 (EC50) (rat) |
| Notes: The smaller the value, the more avidly the drug binds to the site. All proteins are human unless otherwise specified. Footnotes: a = Stimulation of IP1Tooltip inositol phosphate formation. Refs: [12][13][14][15][16][17][18][19][20][21] | |
The interactions of 5-MeO-DALT with various targets have been reported.[14][15][16][20][18] It binds to a variety of serotonin receptors, as well as a number of other targets.[14][15][16][20][22] The drug is a potent full agonist of the serotonin 5-HT1A and 5-HT2A receptors.[16][17][19][18] It is also an agonist of the serotonin 5-HT2B and 5-HT2C receptors.[17]
Uniquely the substance displayed weak agonist-like activity at the μ-opioid (MOR) and δ-opioid receptors (DOR) along with more significant activity at the κ-opioid receptor (KOR) (∼76% of salvinorin A at 1 μM concentration) with G protein bias over β-arrestin activation.[21]
Similarly to other psychedelics, 5-MeO-DALT produces the head-twitch response, a behavioral proxy of psychedelic-like effects, in rodents.[8][16][15] The drug fully substitutes for the serotonergic psychedelic DOM in rodent drug discrimination tests.[23] Conversely, 5-MeO-DALT does not substitute for the entactogen MDMA in such tests.[23] 5-MeO-DALT produces dose-dependent hyperlocomotion in rodents, followed by hypolocomotion at the highest assessed dose.[23][16] This is in contrast to many other psychedelic tryptamines, which tend to produce only hypolocomotion.[23] 5-MeO-DMT and 5-MeO-AMT are locomotor depressants, whereas 5-MeO-DET and 5-MeO-MiPT are mixed locomotor stimulants/depressants similarly to 5-MeO-DALT.[23] It also produces hypothermia.[16]
The head-twitch response induced by 5-MeO-DALT in rodents was found to be positively related to its serotonin 5-HT2A receptor affinity and negatively related to its serotonin 5-HT1A receptor affinity.[15] In relation to this, multiple targets appear to contribute to the effects of 5-MeO-DALT.[15][5]
Pharmacokinetics
The metabolism and cytochrome P450 inhibition of 5-MeO-DALT has been described in scientific literature.[9][24][25][26]
Chemistry
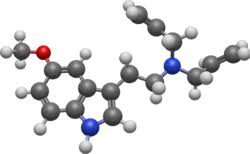
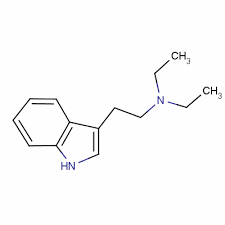
The full name of the chemical is N–allyl–N-[2-(5-methoxy-1H-indol-3-yl)ethyl] prop-2-en-1- amine. It is related to the compounds 5-MeO-DPT, DALT, 4-HO-DALT, and 4-AcO-DALT.
Synthesis
The chemical synthesis of 5-MeO-DALT has been described.[1]
Crystal structure
In April 2020, Chadeayne et al. solved the crystal structure of the freebase form of 5-MeO-DALT.[27]
Analogues
Analogues of 5-MeO-DALT include diallyltryptamine (DALT), 4-HO-DALT (daltocin), 4-AcO-DALT (dalcetin), NB-5-MeO-DALT, 5-MeO-DMT, 5-MeO-DET, 5-MeO-DPT, 5-MeO-DiPT, 5-MeO-MALT, 5-MeO-MiPT, and 5-MeO-iPALT (ASR-3001), among others.
History
The first material regarding the synthesis and effects of 5-MeO-DALT was sent from Alexander Shulgin to a research associate named Murple in May 2004, after which it was circulated online. In June 2004 5-MeO-DALT became available from internet research chemical vendors after being synthesized by commercial laboratories in China. In August 2004 the synthesis and effects of 5-MeO-DALT were published by Erowid.[4] Shulgin has stated that 5-MeO-DALT had not previously existed in the scientific literature.[1] 5-MeO-DALT was not included in the original published version of TiHKAL, but an entry for the compound was subsequently written and released in 2004.[4] The drug was encountered as a novel designer drug by at least 2006.[3][4][5][6]
Society and culture
Legal status
Canada
5-MeO-DALT is not a controlled substance in Canada as of 2025.[28]
China
As of October 2015 5-MeO-DALT is a controlled substance in China.[29]
Japan
5-MeO-DALT became a controlled substance in Japan from April 2007, by amendment to the Pharmaceutical Affairs Law.[30]
Singapore
5-MeO-DALT is listed in the Fifth Schedule of the Misuse of Drugs Act (MDA) and therefore illegal in Singapore as of May 2015.[31]
Sweden
Sveriges riksdag added 5-MeO-DALT to schedule I (“substances, plant materials and fungi which normally do not have medical use”) as narcotics in Sweden as of May 1, 2012, published by Medical Products Agency in their regulation LVFS 2012:6 listed as 5-MeO-DALT N-allyl-N-[2-(5-metoxi-1H-indol-3-yl)etyl]-prop-2-en-1-amin.[32]
United Kingdom
5-MeO-DALT became a Class A drug in the UK on January 7, 2015 after an update to the tryptamine blanket ban.
United States
5-MeO-DALT is not scheduled at the federal level in the United States,[33] but it is likely that it could be considered an analog of 5-Meo-DiPT, which is a controlled substance in USA, or an analog of another tryptamine, in which case purchase, sale, or possession could be prosecuted under the Federal Analog Act.
Florida
5-MeO-DALT is a Schedule I controlled substance in the state of Florida making it illegal to buy, sell, or possess in Florida.[34]
Louisiana
5-MeO-DALT is a Schedule I controlled substance in the state of Louisiana making it illegal to buy, sell, or possess in Louisiana.[35]
Research
Cluster headache
Anecdotal reports[36] and a small-scale trial[37] indicate the potential of 5-MeO-DALT for the treatment of cluster headache, one of the most excruciating conditions known to medicine.[38] These observations are consistent with evidence of efficacy of other chemically-related indoleamines in the treatment of cluster headache.[39]
Reviews
There are no reviews yet.