Buy 5-MeO-DBT Cas 73785-42-9
Buy 5-MeO-DBT Cas 73785-42-9
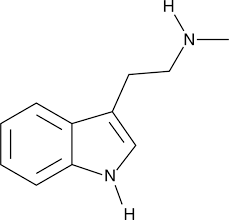
5-MeO-DBT, also known as 5-methoxy-N,N-dibutyltryptamine, is a serotonin receptor modulator, and a rare substituted tryptamine derivative, which is thought to be a psychoactive substance.[1][2]
Unlike many other related compounds it exhibits very low efficacy for the 5-HT2A receptor.[2]
5-MeO-DBT was first described in the literature by Alexander Shulgin in his 1997 book TiHKAL (Tryptamines I Have Known and Loved).[1] It was encountered as a novel designer drug by 2019[3][4] and was assessed pharmacologically in 2023.[2] The drug is controlled under drug analogue legislation in a number of jurisdictions.[5]
Use and effects
In his book TiHKAL (Tryptamines I Have Known and Loved), Alexander Shulgin briefly mentioned 5-MeO-DBT and described it as a known compound with unknown activity.[1] Relatedly, the properties and effects of 5-MeO-DBT are unknown.[1] In any case, related drugs like dibutyltryptamine (DBT) and 4-HO-DPT have been reported to yield disappointing effects.[1][6]
Interactions
Pharmacology
Pharmacodynamics
| Target | Affinity (Ki, nM) |
|---|---|
| 5-HT1A | 337 (Ki) 267 (EC50Tooltip half-maximal effective concentration) 106% (EmaxTooltip maximal efficacy) |
| 5-HT2A | 562 (Ki) 620a (EC50) 18%a (Emax) |
| 5-HT2C | 3,130 (Ki) 2,400a (EC50) 76%a (Emax) |
| SERTTooltip Serotonin transporter | 1,180 (Ki) 2,120 (IC50) |
| Notes: The smaller the value, the more avidly the drug interacts with the site. Footnotes: a = Stimulation of IP1Tooltip inositol phosphate formation. Sources: [2] | |
Based on limited evidence, 5-MeO-DBT acts as a non-selective serotonin receptor agonist with the highest potency and efficacy at the 5-HT1A receptor.[2] It has a similar potency to 5-MeO-MiPT for this target.[2] The substance, unlike many other substituted tryptamines, acts as a very weak and low efficacy partial agonist for the 5-HT2A receptor.[2] Among the group of related tryptamine analogues it also displayed the lowest efficacy for the 5-HT2C receptor.[2]
5-MeO-DBT decreased locomotor activity and failed to substitute for the discriminative stimulus effects of DOM in rodent drug discrimination tests.[6]
Chemistry
Analogues
Analogues of 5-MeO-DBT include dibutyltryptamine (DBT), 4-HO-DBT, 5-MeO-DMT, 5-MeO-DET, 5-MeO-DPT, 5-MeO-DiPT, 5-MeO-DALT, 5-MeO-DsBT, and 5-MeO-MBT, among others.[1]
History
5-MeO-DBT was first described in the literature by Alexander Shulgin in his 1997 book TiHKAL (Tryptamines I Have Known and Loved).[1] It was encountered as a novel designer drug by 2019.[3][4]
Society and culture
Legal status
United States
Alabama
5-MeO-DBT was made schedule I at the state level in Alabama on September 13, 2024.[5]
Reviews
There are no reviews yet.