Buy 5-Bromo-DMT Cas 17274-65-6
Buy 5-Bromo-DMT Cas 17274-65-6
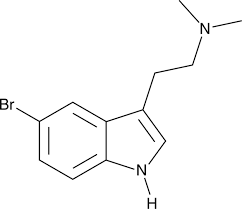
5-Bromo-DMT, or 5-Br-DMT, also known as 5-bromo-N,N-dimethyltryptamine or by informal names like sea DMT or SpongeBob DMT[dubious – discuss], is a psychedelic drug and brominated indole alkaloid of the tryptamine family related to dimethyltryptamine (DMT).[1][2][3] It is the 5-bromo derivative of DMT.[1] The drug is naturally occurring in the sponges Smenospongia aurea and Smenospongia echina, as well as in Verongula rigida (0.00142% dry weight) alongside 5,6-dibromo-DMT (0.35% dry weight) and seven other alkaloids.[1][4][5][6][7] It has been encountered as a novel designer drug.[8][9]
Use and effects
5-Bromo-DMT was only briefly mentioned in Alexander Shulgin‘s book TiHKAL (Tryptamines I Have Known and Loved) and its properties and effects were not described.[10] Subsequently, the drug has been reported by others to have a dose of 20 to 50 mg smoked and a duration of 15 minutes to 1.5 hours.[1][8] It is minimally active or inactive orally.[1] It was described as producing mild psychedelic effects, such as visuals, pronounced tactile effects, and euphoria.[1][8] 5-Bromo-DMT was said to be similar to low-dose DMT, but also distinct from it.[1] A 50 mg dose was said to be near the limit of what can be physically inhaled.[1] However, it was thought that greater exposure to the drug nonetheless might be able to produce stronger effects.[1] These findings were reported in a Shulgin- or TiHKAL-like style via credible anonymous personal communication with Hamilton Morris and Jason Wallach.[1]
Interactions
Pharmacology
Pharmacodynamics
5-Bromo-DMT is a partial agonist of the serotonin 5-HT2A receptor, with an affinity (Ki) of 138 nM, an EC50Tooltip half-maximal effective concentration of 77.7 to 3,090 nM, and an EmaxTooltip maximal efficacy of 34 to 100%.[2][3][11] It also shows affinity for the serotonin 5-HT1A, 5-HT2B, and 5-HT2C receptors and for the serotonin transporter (SERT) (Ki = 16.9 nM, 403 nM, 193 nM, and 971 nM, respectively).[3] The drug is a weak serotonin 5-HT1A receptor full agonist (EC50 = 1,810 nM; Emax = 94%) and a very weak serotonin reuptake inhibitor (IC50Tooltip half-maximal inhibitory concentration = 8,055 nM).[3]
In contrast to 5-fluoro-DMT and 5-chloro-DMT, 5-bromo-DMT failed to significantly produce the head-twitch response, a behavioral proxy of psychedelic effects, in rodents.[2][3][12][13] As such, 5-bromo-DMT would be expected to be non-hallucinogenic in humans.[2] In addition, 5-bromo-DMT antagonized the head-twitch response induced by 5-fluoro-DMT.[3] On the other hand, 5-bromo-DMT produced antidepressant-like effects, hypolocomotion or sedative-like effects, and hypothermia in rodents.[3][2][1][14] Moreover, 5-bromo-DMT has been found to produce psychoplastogenic effects.[3]
Chemistry
Synthesis
The chemical synthesis of 5-bromo-DMT has been described.[1]
Analogues
Analogues of 5-bromo-DMT include 5,6-dibromo-DMT, 5-fluoro-DMT, 5-chloro-DMT, bretisilocin (5-fluoro-MET), 5-fluoro-DET, 5-fluoro-AMT, 5-chloro-AMT, BK-5Br-NM-AMT, 5-nitro-DMT, convolutindole A, desformylflustrabromine, and plakohypaphorine, among others.
History
5-Bromo-DMT was briefly mentioned by Alexander Shulgin in his 1991 book TiHKAL (Tryptamines I Have Known and Loved), but he did not synthesize or test it.[1][10] Hamilton Morris and Jason Wallach reported the properties and hallucinogenic effects of 5-bromo-DMT in humans in 2013 via publication of credible personal communication with an anonymous “Dr. Osculum”.[1] 5-Bromo-DMT was described as a novel designer drug by 2020.[8][9]
Society and culture
Legal status
Canada
5-Bromo-DMT is not a controlled substance in Canada as of 2025.[15]
Singapore
5-Bromo-DMT is specifically listed as a controlled drug in Singapore.[16]
United States
5-Bromo-DMT is not an explicitly controlled substance in the United States.[17] However, it could be considered a controlled substance under the Federal Analogue Act if intended for human consumption.

Reviews
There are no reviews yet.