Buy MEM Cas 16128-88-4
Buy MEM Cas 16128-88-4
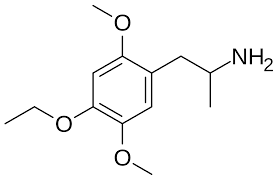
MEM, also known as 2,5-dimethoxy-4-ethoxyamphetamine or as TMA2-4-EtO, is a psychedelic drug of the phenethylamine, amphetamine, and DOx families related to TMA-2.[1] It is the analogue of TMA-2 in which the methoxy group at the 4 position has been replaced with an ethoxy group.[1] The drug was first described in the scientific literature by Alexander Shulgin by 1968.[2]
Use and effects
In his book PiHKAL (Phenethylamines I Have Known and Loved), Alexander Shulgin lists MEM’s dose as 20 to 50 mg orally and its duration as 10 to 14 hours.[1][3] Its effects have been reported to include color enhancement, visual phenomena, and pattern movement, among others.[1]
Interactions
Pharmacology
Pharmacodynamics
| Target | Affinity (Ki, nM) |
|---|---|
| 5-HT1A | >10,000 |
| 5-HT1B | >10,000 |
| 5-HT1D | >10,000 |
| 5-HT1E | >10,000 |
| 5-HT1F | ND |
| 5-HT2A | 73.0–3,948 (Ki) 47.5–295 (EC50Tooltip half-maximal effective concentration) 88–105% (EmaxTooltip maximal efficacy) |
| 5-HT2B | 64.5–763 (Ki) 437–557 (EC50) 70–96% (Emax) |
| 5-HT2C | 124–>10,000 (Ki) 29.9–248 (EC50) 98–129% (Emax) |
| 5-HT3 | >10,000 |
| 5-HT4 | ND |
| 5-HT5A | >10,000 |
| 5-HT6 | >10,000 |
| 5-HT7 | 7,156 |
| α1A, α1B | >10,000 |
| α1D | ND |
| α2A–α2C | >10,000 |
| β1, β2 | >10,000 |
| β3 | ND |
| D1–D5 | >10,000 |
| H1–H4 | >10,000 |
| M1–M5 | >10,000 |
| I1 | >10,000 |
| σ1 | 5,077 |
| σ2 | >10,000 |
| TAAR1Tooltip Trace amine-associated receptor 1 | ND |
| SERTTooltip Serotonin transporter | >10,000 (Ki) |
| NETTooltip Norepinephrine transporter | >10,000 (Ki) |
| DATTooltip Dopamine transporter | >10,000 (Ki) |
| Notes: The smaller the value, the more avidly the drug binds to the site. All proteins are human unless otherwise specified. Refs: [4][5][6][7][8][9][10] | |
MEM is a serotonergic psychedelic and acts as a selective serotonin 5-HT2 receptor agonist.[6][7][8][9][10] It is specifically a full agonist of the serotonin 5-HT2A and 5-HT2C receptors and to a lesser extent is a partial to full agonist of the serotonin 5-HT2B receptor.[6][7][9] The psychedelic effects of MEM are thought to be mediated by serotonin 5-HT2A receptor activation.[7]
Chemistry
MEM, also known as 2,5-dimethoxy-4-ethoxyamphetamine, is a phenethylamine, amphetamine, and DOx derivative. It is the analogue and derivative of 2,4,5-trimethoxyamphetamine (TMA-2) in which a 4-ethoxy group is present instead of a 4-methoxy group.
Synthesis
The chemical synthesis of MEM has been described.[1]
Derivatives
A variety of derivatives of MEM have been developed and studied, not only by Alexander Shulgin but also by for instance Daniel Trachsel and colleagues.[11][12] These include MPM, MIPM, MALM, MBM, MAM, MMALM, MFEM, MDFEM, and MTFEM, among others.[11][12][13]
History
MEM was first synthesized by Alexander Shulgin.[1][2] It was first described by him in the scientific literature by 1968.[2] Subsequently, Shulgin described MEM in greater detail in his 1991 book PiHKAL (Phenethylamines I Have Known and Loved).[1]
Society and culture
Legal status
Canada
MEM is a controlled substance in Canada.[14]
United States
MEM is not an explicitly controlled substance in the United States.[15] However, it could be considered a controlled substance under the Federal Analogue Act if intended for human consumption.
Reviews
There are no reviews yet.