Buy 2C-N Cas 261789-00-8
Buy 2C-N Cas 261789-00-8
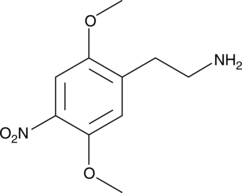
2C-N, also known as 4-nitro-2,5-dimethoxyphenethylamine, is a psychedelic drug of the phenethylamine and 2C families.[1] It is taken orally.[1]
2C-N was first synthesized by Alexander Shulgin and was described in his 1991 book PiHKAL (Phenethylamines I Have Known and Loved).[1]
Use and effects
In his book PiHKAL (Phenethylamines I Have Known and Loved), Alexander Shulgin lists 2C-N’s dose range as 100 to 150 mg or more orally and a duration of 4 to 6 hours.[1][2] It has an estimated typical dose of about 120 mg orally.[2] Its onset is within 30 minutes and peak effects occur after 1 hour.[1] The effects of 2C-N have been reported to include some visual changes, similarities to MDMA, lightheadedness, eye wiggling, easier conversation, and improved mood.[1] It was described as a “strange material, but okay”.[1]
Interactions
2C drugs like 2C-N are known to be metabolized by the monoamine oxidase (MAO) enzymes MAO-A and MAO-B.[3][4] Monoamine oxidase inhibitors (MAOIs) such as phenelzine, tranylcypromine, moclobemide, and selegiline may potentiate the effects of 2C drugs like 2C-N.[3][4][5] This may result in overdose and serious toxicity.[5][3]
Pharmacology
Pharmacodynamics
| Target | Affinity (Ki, nM) |
|---|---|
| 5-HT1A | 1,450–2,200 |
| 5-HT1B | >10,000 |
| 5-HT1D | 832 |
| 5-HT1E | 676 |
| 5-HT1F | ND |
| 5-HT2A | 23.5–72.4 (Ki) 32.2–170 (EC50Tooltip half-maximal effective concentration) 48–100% (EmaxTooltip maximal efficacy) |
| 5-HT2B | 123 (Ki) 730 (EC50) 74% (Emax) |
| 5-HT2C | 162–370 (Ki) ND (EC50) ND (Emax) |
| 5-HT3 | >10,000 |
| 5-HT4 | ND |
| 5-HT5A | >10,000 |
| 5-HT6 | 251 |
| 5-HT7 | >10,000 |
| α1A | >15,000 |
| α1B, α1D | >10,000 |
| α2A | 240–1,300 |
| α2B | 2,240 |
| α2C | 891 |
| β1–β3 | >10,000 |
| D1 | 19,000 |
| D2 | 6,100–>10,000 |
| D3 | 20,000 |
| D4, D5 | >10,000 |
| H1 | >25,000 |
| H2 | >10,000 |
| H3 | 5,500 |
| H4 | >10,000 |
| M1–M5 | >10,000 |
| I1 | ND |
| σ1, σ2 | >10,000 |
| ORs | >10,000 |
| TAAR1Tooltip Trace amine-associated receptor 1 | >20,000 (Ki) (mouse) 340 (Ki) (rat) 15,000 (EC50) (mouse) 250 (EC50) (rat) >10,000 (EC50) (human) 28% (Emax) (mouse) 59% (Emax) (rat) |
| SERTTooltip Serotonin transporter | 32,000 (Ki) 154,000 (IC50Tooltip half-maximal inhibitory concentration) ND (EC50) |
| NETTooltip Norepinephrine transporter | >30,000 (Ki) 287,000 (IC50) ND (EC50) |
| DATTooltip Dopamine transporter | >30,000 (Ki) >900,000 (IC50) ND (EC50) |
| MAO-ATooltip Monoamine oxidase A | ND (IC50) |
| MAO-BTooltip Monoamine oxidase B | 66,000 (IC50) |
| Notes: The smaller the value, the more avidly the drug binds to the site. All proteins are human unless otherwise specified. Refs: [6][7][8][9][10][11][12] | |
2C-N is a low-potency partial agonist of the serotonin 5-HT2A, 5-HT2B, and 5-HT2C receptors.[7][9][13][11]
Chemistry
Properties
Salts of 2C-N have a bright yellow to orange color due to the presence of the nitro group,[citation needed] unlike all other members of the 2C family in which the salts are white.
Synthesis
The chemical synthesis of 2C-N has been described.[1] It is synthesized by the mixed acid nitration of 2C-H using sulfuric acid and nitric acid.[1]
Analogues
Analogues of 2C-N include DON, 2C-CN, and 25N-NBOMe, among others.
History
2C-N was first described in the scientific literature by at least 1991.[1]
Society and culture
Legal status
Canada
As of October 31, 2016, 2C-N is a controlled substance (Schedule III) in Canada.[14]
United Kingdom
2C-N and most (possibly all) other compounds featured in PiHKAL are illegal drugs in the United Kingdom.
United States
In the United States, 2C-N is a Schedule 1 controlled substance.[15]
Reviews
There are no reviews yet.