Buy 2C-T-4 Cas 207740-25-8
Buy 2C-T-4 Cas 207740-25-8
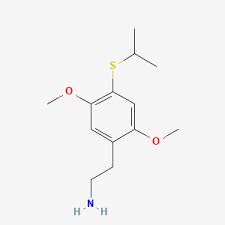
2C-T-4, also known as 4-isopropylthio-2,5-dimethoxyphenethylamine, is a psychedelic drug of the phenethylamine and 2C families.[1][2] It is taken orally.[1]
2C-T-4 was first described in the scientific literature by Alexander Shulgin and colleagues in 1991.[3] Shortly after this, Shulgin described 2C-T-17 in greater detail in his 1991 book PiHKAL (Phenethylamines I Have Known and Loved).[1]
Use and effects
In his book PiHKAL (Phenethylamines I Have Known and Loved), Alexander Shulgin lists 2C-T-4’s dose as 8 to 20 mg orally and its duration as 12 to 18 hours.[1] Its onset is 30 minutes to 2 hours and peak effects occur after 3 hours.[1] The effects of 2C-T-4 have been described and include psychedelic visuals among others.[1] Shulgin devoted a chapter in the first part of PiHKAL to 2C-T-4, describing an intense “plus-four” experience on the Shulgin Rating Scale with a 12 mg dose.[1]
Interactions
2C drugs are metabolized by the monoamine oxidase (MAO) enzymes MAO-A and MAO-B.[4][5] Monoamine oxidase inhibitors (MAOIs) such as phenelzine, tranylcypromine, moclobemide, and selegiline may potentiate the effects of 2C drugs.[4][5][6] This may result in overdose and serious toxicity.[6][4]
Pharmacology
Pharmacodynamics
| Target | Affinity (Ki, nM) |
|---|---|
| 5-HT1A | 470–916 |
| 5-HT1B | ND |
| 5-HT1D | ND |
| 5-HT1E | ND |
| 5-HT1F | ND |
| 5-HT2A | 27.9–54 (Ki) 5.5–220 (EC50Tooltip half-maximal effective concentration) 56–87% (EmaxTooltip maximal efficacy) |
| 5-HT2B | ND (Ki) 63–160 (EC50) 68–75% (Emax) |
| 5-HT2C | 180–295 (Ki) ND (EC50) ND (Emax) |
| 5-HT3 | ND |
| 5-HT4 | ND |
| 5-HT5A | ND |
| 5-HT6 | ND |
| 5-HT7 | ND |
| α1A | 11,000 |
| α1B, α1D | ND |
| α2A | 130–217 |
| α2B, α2C | ND |
| β1–β3 | ND |
| D1 | 20,000 |
| D2 | 16,000 |
| D3 | 19,000 |
| D4, D5 | ND |
| H1 | >25,000 |
| H2–H4 | ND |
| M1–M5 | ND |
| I1 | ND |
| σ1, σ2 | ND |
| TAAR1Tooltip Trace amine-associated receptor 1 | 2,337–4,500 (Ki) (mouse) 19–53 (Ki) (rat) 3,700 (EC50) (mouse) 83 (EC50) (rat) >30,000 (EC50) (human) 51% (Emax) (mouse) 67% (Emax) (rat) |
| SERTTooltip Serotonin transporter | >30,000 (Ki) 113,000 (IC50Tooltip half-maximal inhibitory concentration) ND (EC50) |
| NETTooltip Norepinephrine transporter | 17,000 (Ki) 134,000 (IC50) ND (EC50) |
| DATTooltip Dopamine transporter | >30,000 (Ki) 294,000 (IC50) ND (EC50) |
| Notes: The smaller the value, the more avidly the drug binds to the site. All proteins are human unless otherwise specified. Refs: [7][8][9][10] | |
2C-T-4 acts as a serotonin 5-HT2 receptor agonist, including of the serotonin 5-HT2A receptor.[9][8] The mechanism that produces 2C-T-4’s hallucinogenic effects has not been specifically established, however it is most likely to result from 5-HT2A receptor activation in the brain, a mechanism of action shared by all of the hallucinogenic tryptamines and phenethylamines for which the mechanism of action is known.
Chemistry
2C-T-4 is the 2-carbon homologue of Aleph-4.[1] The full chemical name is 2-[4-(isopropylthio)-2,5-dimethoxyphenyl]-ethanamine.[1] The drug has structural and pharmacodynamic properties similar to 2C-T-7 and 2C-T-19.[1] A notable analogue of 2C-T-4 is the Ψ-PEA compound Ψ-2C-T-4.[1]
Synthesis
The chemical synthesis of 2C-T-4 has been described.[1]
Analogues
Analogues of 2C-T-4 include 2C-T, 2C-T-2, 2C-T-7, and Aleph-4, among others.[1]
History
2C-T-4 was first described in the scientific literature by Alexander Shulgin and colleagues in a journal article in 1991.[3] Shortly thereafter, it was described in greater detail by Shulgin in his 1991 book PiHKAL (Phenethylamines I Have Known and Loved).[1]
Society and culture
Legal status
Canada
As of October 31, 2016, 2C-T-4 is a controlled substance (Schedule III) in Canada.[11]
China
As of October 2015 2C-T-4 is a controlled substance in China.[12]
Denmark
2C-T-4 is added to the list of Schedule B controlled substances.[13]
Sweden
Sveriges riksdags health ministry Statens folkhälsoinstitut classified 2C-T-4 as “health hazard” under the act Lagen om förbud mot vissa hälsofarliga varor (translated Act on the Prohibition of Certain Goods Dangerous to Health) as of Jul 15, 2007, in their regulation SFS 2007:600 listed as 2,5-dimetoxi-4-isopropyltiofenetylamin (2C-T-4), making it illegal to sell or possess.[14]
United States
As of July 9, 2012, 2C-T-4 is a Schedule I substance in the United States, under the Synthetic Drug Abuse Prevention Act of 2012.[15]
Reviews
There are no reviews yet.