Buy 4-Acetoxy-N (N-dimethyltryptamine) Cas 92292-84-7
Buy 4-Acetoxy-N (N-dimethyltryptamine) Cas 92292-84-7
4-Acetoxy-N,N-dimethyltryptamine (4-AcO-DMT or 4-acetoxy-DMT), also known as O-acetylpsilocin or psilacetin, is a psychedelic drug of the tryptamine family related to psilocybin and psilocin.[4][5][6][7] It is a synthetic derivative of psilocin (4-HO-DMT) in which the hydroxyl group has been acetylated, and is the analogue of psilocybin (4-PO-DMT) in which the phosphate ester has been replaced with an acetate ester.[4][5][6] The drug is a prodrug of psilocin and is used orally similarly to psilocybin.[4][5][8][1][2]
As a prodrug of psilocin, 4-AcO-DMT acts as a non-selective serotonin receptor agonist, including of the serotonin 5-HT2A receptor.[4][9] The hallucinogenic effects of psilocin are thought to be mediated by activation of this receptor, although other receptors also contribute to its effects.[10][11][4] 4-AcO-DMT’s effects are reported to be similar to those of psilocybin and psilocybin mushrooms.[5][8][4] However, it has been said to have reduced side effects such as nausea and body load that can be caused by ingestion of whole psilocybin mushrooms.[5][8][4] It is also said to have a faster onset and shorter duration than psilocybin.[8] The drug is not expected to differ from psilocybin or psilocin in terms of safety.[4] 4-AcO-DMT is modestly less potent by weight than psilocybin in animals when they are given at equimolar doses.[5]
4-AcO-DMT was first described in a patent by Albert Hofmann in 1963 and its chemical synthesis was improved by David E. Nichols and colleagues in 1999.[5][9][6] It was suggested by Nichols as a more economical and accessible alternative to psilocybin for use in scientific research, as the synthesis of psilocybin is more challenging and as psilocybin is a controlled substance.[5][9][6] 4-AcO-DMT was first detected as a designer drug in Europe in 2009.[9] It became increasingly prevalent as a recreational drug in the 2010s and has been the most commonly used novel tryptamine.[5][8] In the 2020s, 4-AcO-DMT became widely encountered in the form of mushroom edibles in the United States as an alternative to psilocybin.[12][13][14][15] Relatedly, it has sometimes been referred to as “synthetic shrooms“.[7] Mushrooms edibles may contain 4-AcO-DMT, Amanita muscaria mushroom constituents, or non-mushroom drugs such as bath salts, and have been linked to poisonings and deaths.[16][7][15][12]
4-AcO-DMT is not scheduled under United States law or any international drug schedules, including the United Nations 1971 Convention on Psychotropic Substances, making it a potentially more accessible alternative to psilocybin for research.[5] It can be imported and possessed for research in the United States if labeled “not for human consumption,” but using it in vivo is illegal and violates the Federal Analogue Act.[4]
Use and effects
In his book TiHKAL (Tryptamines I Have Known and Loved), Alexander Shulgin lists the same dose range of 10 to 20 mg orally and duration of 3 to 6 hours for psilocin, psilocybin, and 4-AcO-DMT.[1] Another publication gave a 4-AcO-DMT dose range of 10 to 15 mg orally, with a typical dose of 12.5 mg orally, and a duration of about 5 to 8 hours.[2] A further source gave a dose range for the drug of 5 to 30 mg orally, an onset of 15 to 40 minutes, and a duration of 4 to 7 hours.[3] 4-AcO-DMT is a prodrug of psilocin similarly to psilocybin and its effects are reported to be similar or identical to those of psilocybin and psilocybin-containing mushrooms.[5][8][3] However, it is said to produce less nausea and body load than psilocybin-containing mushrooms.[5][8][4] The drug is also often described as having a faster onset and shorter duration than psilocybin.[8] 4-AcO-DMT is modestly less potent by weight than psilocybin in animals when they are given at equimolar doses.[5]
Specific effects of 4-AcO-DMT have been reported to include psychedelic visuals, closed-eye imagery, synesthesia, insights, disembodiment, euphoria, feelings of bliss and unity, oceanic boundlessness, ego dissolution, sedation, cognitive impairment, and spiritual experiences, among others.[2][3] Adverse effects have been reported to include psychological side effects such as anxiety, paranoia, and low mood as well as gastrointestinal side effects such as nausea and vomiting.[2]
Contraindications
Side effects
4-AcO-DMT, as a prodrug of psilocin, is not expected to differ from psilocybin or psilocin in terms of safety.[4]
Interactions
Pharmacology
Pharmacodynamics
| Target | Affinity (Ki, nM) |
|---|---|
| 5-HT1A | 49–567 (Ki) 130–>3,160 (EC50Tooltip half-maximal effective concentration) 0.7%–96% (EmaxTooltip maximal efficacy) |
| 5-HT1B | 31–305 |
| 5-HT1D | 19–36 |
| 5-HT1E | 44–52 |
| 5-HT1F | ND |
| 5-HT2A | 6.0–340 (Ki) 2.4–3,836 (EC50) 16–98% (Emax) |
| 5-HT2B | 4.6–410 (Ki) 2.4–>20,000 (EC50) 1.4–84% (Emax) |
| 5-HT2C | 10–141 (Ki) 9.1–30 (EC50) 86–95% (Emax) |
| 5-HT3 | >10,000 |
| 5-HT4 | ND |
| 5-HT5A | 70–84 |
| 5-HT6 | 57–72 |
| 5-HT7 | 3.5–72 |
| α1A–α1B | >10,000 |
| α2A | 1,379–2,044 |
| α2B | 1,271–1,894 |
| α2C | 4,404 |
| β1–β2 | >10,000 |
| D1 | 20–>14,000 |
| D2 | 3,700–>10,000 |
| D3 | 101–8,900 |
| D4 | >10,000 |
| D5 | >10,000 |
| H1 | 1,600–>10,000 |
| H2–H4 | >10,000 |
| M1–M5 | >10,000 |
| σ1 | >10,000 |
| σ2 | >10,000 |
| I2 | 792 |
| TAAR1 | 1,400 (Ki) (rat) 17,000 (Ki) (mouse) 920–2,700 (EC50) (rodent) >30,000 (EC50) (human) |
| SERTTooltip Serotonin transporter | 3,650–>10,000 (Ki) 662–3,900 (IC50Tooltip half-maximal inhibitory concentration) 561 (EC50) 54% (Emax) |
| NETTooltip Norepinephrine transporter | 13,000 (Ki) 14,000 (IC50) >10,000 (EC50) |
| DATTooltip Dopamine transporter | 6,000–>30,000 (Ki) >100,000 (IC50) >10,000 (EC50) |
| Notes: The smaller the value, the more avidly psilocin interacts with the site. Sources: [17][18][19][20][21][22][23][24][25][26] |
|
4-AcO-DMT is a prodrug of psilocin (4-HO-DMT).[5] As a prodrug of psilocin, 4-AcO-DMT acts as a non-selective agonist of serotonin receptors, including of the serotonin 5-HT2A receptor.[4] The psychedelic effects of 4-AcO-DMT are mediated specifically by activation of the serotonin 5-HT2A receptor.[4]
Similarly to psilocybin, psilocin, and other serotonergic psychedelics, 4-AcO-DMT produces the head-twitch response, a behavioral proxy of psychedelic effects, in rodents.[5][9][7][36] In addition, like psilocybin and other psychedelics, 4-AcO-DMT fully substitutes for the psychedelic DOM in rodent drug discrimination tests.[37] 4-AcO-DMT produces effects such as hypolocomotion and hypothermia in rodents as with psilocin as well.[5]
Pharmacokinetics
There are no clinical studies of the pharmacokinetics of 4-AcO-DMT as of 2024.[5] However, the pharmacokinetics of 4-AcO-DMT have been studied in rodents.[5] The drug was confirmed to act as a prodrug of psilocin similarly to psilocybin (4-PO-DMT).[5] However, given by intraperitoneal injection at equimolar doses, 4-AcO-DMT showed only 70% of the relative bioavailability or total exposure of psilocybin.[5] Hence, 4-AcO-DMT results in modestly lower psilocin levels than psilocybin even when the drugs are given at equivalent doses with adjustment for differences in molecular weight.[5] Along similar lines, the psilocin concentrations with 4-AcO-DMT 15 minutes after administration were 75 to 90% of those of an equimolar dose of psilocybin.[5] The elimination half-life of psilocin was approximately 30 minutes and did not differ between 4-AcO-DMT and psilocybin.[5] Psilocin ester prodrugs like 4-AcO-DMT are cleaved into psilocin by esterase enzymes.[38]
A 2025 in-vitro study examined the stability and metabolism of several psilocin ester prodrugs, including 4-AcO-DMT.[39] The results showed that 4-AcO-DMT was rapidly broken down into psilocin by esterase enzymes, with over 99.9% of the prodrug converted within 5 minutes under conditions mimicking the human body (i.e., in human plasma).[39] These findings support the idea that 4-AcO-DMT is quickly and efficiently converted into psilocin before it enters the bloodstream, and that the prodrug itself likely contributes little to the overall pharmacological effect.[39]
Chemistry

Synthesis
4-AcO-DMT can be obtained by acetylation of psilocin under alkaline or strongly acidic conditions. It is, therefore, a synthetic compound. 4-AcO-DMT is more resistant than psilocin to oxidation under basic conditions due to its acetoxy group. It is not as difficult as psilocybin to synthesize.[citation needed]
Stability
Given enough time in unfavorable conditions, 4-AcO-DMT can sometimes turn into a degraded form which is brown in color and can even progress into a brown/black tar-like substance. Researchers hypothesize this is a polymerization reaction and is said to have no effect on the potency of the substance. Preliminary GCMS analysis of the closely related homologue 4-AcO-DET suggests that this degraded form of 4-AcO-DMT consists mainly of the hydroxy form of the parent molecule.[40]
Analogues
4-AcO-DMT is closely related to psilocin (4-HO-DMT) and psilocybin (4-PO-DMT).[1] It is a lower homologue of 4-AcO-MET, 4-AcO-DET, 4-AcO-MiPT, and 4-AcO-DiPT.[1] Other analogues of 4-AcO-DMT include 4-AcO-DPT, 4-MeO-DMT, and 4-PrO-DMT (O-propionylpsilocin).[1][41] Other related prodrugs of psilocin besides 4-AcO-DMT, 4-PrO-DMT, and psilocybin include CT-4201, EB-002, RE-109 (4-GO-DMT), and MSP-1014.
History
4-AcO-DMT and several other esters of psilocin were patented on January 16, 1963 by Sandoz via Albert Hofmann and Franz Troxler.[5][9][42][43] The drug’s chemical synthesis was improved by David E. Nichols and colleagues in 1999 and it was suggested as a more economical and accessible alternative to psilocybin for use in scientific research.[5][9][6] 4-AcO-DMT was first detected as a designer drug in Europe in 2009.[9] It became increasingly prevalent as a recreational drug in the 2010s and has been the most commonly used novel tryptamine.[5][8] In the 2020s, 4-AcO-DMT became widely encountered in the form of mushroom edibles in the United States as an alternative to psilocybin and psilocybin-containing mushrooms.[12][13][14][15]
Society and culture
Legal status
International
4-AcO-DMT is not scheduled under any international drug schedules, including the United Nations 1971 Convention on Psychotropic Substances, making it a potentially more accessible alternative to psilocybin for research.[44]
Australia
4-AcO-DMT can be considered an analog of psilocin making it a Schedule 9 prohibited substance in Australia under the Poisons Standard (October 2015).[45] A Schedule 9 substance is a substance which may be abused or misused, the manufacture, possession, sale or use of which should be prohibited by law except when required for medical or scientific research, or for analytical, teaching or training purposes with approval of Commonwealth and/or State or Territory Health Authorities.[45]
Canada
4-AcO-DMT is not a controlled substance in Canada as of 2025.[46]
Czech Republic
4-AcO-DMT is prohibited in Czech Republic except strictly limited research and therapeutical purposes.[47]
Germany
4-AcO-DMT is banned in Germany according to the BtMG since it is an ester of psilocin.[48]
Israel
4-AcO-DMT is technically illegal in Israel as of being a derivative of dimethyltryptamine (DMT).[citation needed]
Italy
4-AcO-DMT is illegal in Italy as it is an ester of a prohibited substance.[citation needed]
Sweden
The Riksdag added 4-AcO-DMT to Narcotic Drugs Punishments Act under swedish schedule I (“substances, plant materials and fungi which normally do not have medical use” ) as of January 25, 2017, published by Medical Products Agency (MPA) in regulation HSLF-FS 2017:1 listed as “4-acetoxi-N,N-dimetyltryptamin”.[49]
United Kingdom
4-AcO-DMT, being an ester of psilocin, is a Class A drug in the United Kingdom under the Misuse of Drugs Act 1971.[50]
United States
4-AcO-DMT is not an explicitly controlled substance in the United States.[51][4][52] However, it may be considered an analogue of psilocin and psilocybin under the Federal Analogue Act, but only if intended or used for human consumption.[4][52] Conversely, if not intended for human consumption, for instance if used only for research purposes, it may be considered legal.[4][52]
While not controlled at the federal level, 4-AcO-DMT is listed as a controlled substance at the state level in multiple states in the United States, including in Alabama which has made it a schedule I at the state level on March 18, 2014, along with several other
4-Acetoxy-N,N-dimethyltryptamine
Content Navigation
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
Synonyms
Canonical SMILES
4-Acetoxy-N,N-dimethyltryptamine, commonly referred to as 4-AcO-DMT or psilacetin, is a semi-synthetic psychedelic compound belonging to the tryptamine class. It is structurally related to psilocybin and psilocin, the active compounds found in certain mushrooms. The compound features an acetoxy group attached to the nitrogen atom of the tryptamine backbone, which differentiates it from its natural counterparts. It is believed that 4-AcO-DMT acts as a prodrug, converting into psilocin upon ingestion, thereby producing psychedelic effects similar to those of psilocybin .
The mechanism of action of 4-AcO-DMT is similar to psilocin. Once converted to psilocin, it acts as a serotonin 2A (5-HT2A) receptor agonist in the brain []. This means it mimics the effects of serotonin, a neurotransmitter involved in mood, perception, and cognition. The activation of 5-HT2A receptors is believed to be responsible for the hallucinogenic effects of 4-AcO-DMT [].
The primary reaction involving 4-AcO-DMT occurs during its metabolism in the human body. Upon ingestion, the acetoxy group is cleaved by deacetylases and acetyltransferases during first-pass metabolism, converting it into psilocin, which is responsible for its psychoactive effects. Additionally, 4-AcO-DMT can undergo degradation under unfavorable conditions, resulting in a brown tar-like substance without affecting potency .
4-AcO-DMT exhibits significant biological activity primarily through its interaction with serotonin receptors, particularly the 5-HT2A receptor. This interaction is thought to be responsible for its psychedelic effects, which can include euphoria, altered perception of time and space, and profound introspective insights. Users have reported emotional states ranging from blissful euphoria to anxiety-inducing experiences .
Preliminary studies suggest that 4-AcO-DMT may also have therapeutic potential for mental health conditions, including addiction. Animal studies have indicated that it can prevent and reverse heroin and nicotine addictions by modulating brain-derived neurotrophic factor levels .
The synthesis of 4-AcO-DMT typically involves the acetylation of psilocin or other tryptamine precursors. The process requires advanced knowledge of organic chemistry and access to specific reagents. Common methods include:
4-AcO-DMT has garnered interest in both recreational and therapeutic contexts. As a research chemical, it serves as a potential alternative to psilocybin in pharmacological studies due to its lower cost and ease of synthesis. Its psychoactive properties make it a subject of interest in exploring psychedelic therapy for mental health issues such as depression and anxiety .
4-AcO-DMT shares structural similarities with several other compounds in the tryptamine class. Below are some notable comparisons:
| Compound Name | Structural Features | Unique Characteristics |
|---|---|---|
| Psilocybin | O-phosphoryloxy group | Naturally occurring in magic mushrooms |
| Psilocin | Hydroxy group | Active form produced from psilocybin |
| N,N-Dimethyltryptamine | Two methyl groups on nitrogen | Endogenous compound with strong psychoactive effects |
| 4-Acetoxy-N,N-diethyltryptamine | Ethyl groups instead of methyl groups | Different psychoactive profile |
| 4-Hydroxy-N,N-dimethyltryptamine | Hydroxy group instead of acetoxy | More studied for therapeutic uses |
4-AcO-DMT’s unique acetoxy group provides distinct pharmacological properties compared to these compounds while retaining the core tryptamine structure that underpins their similar psychedelic effects .
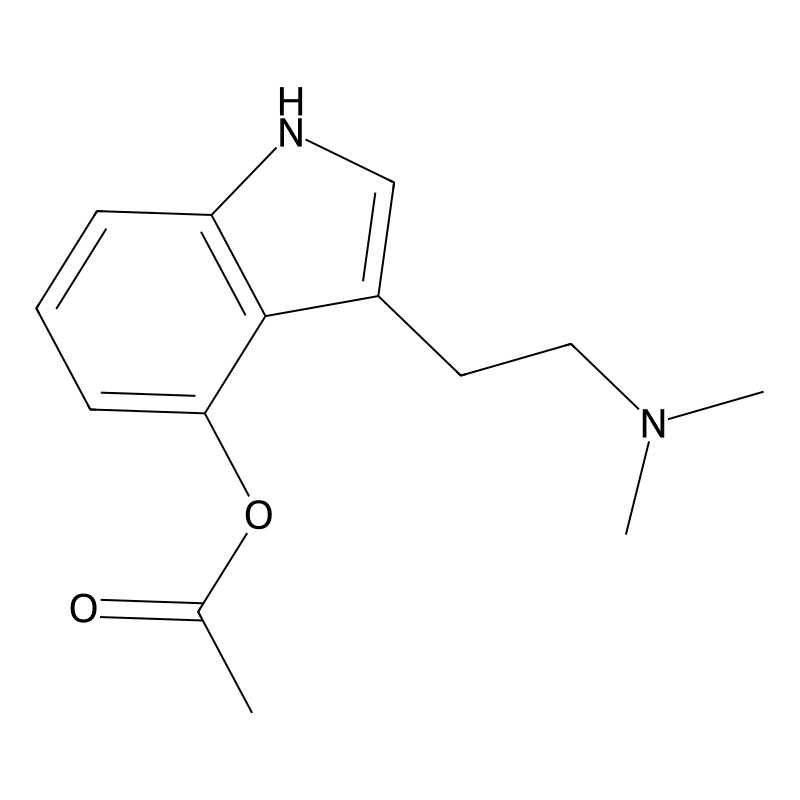
4-Acetoxy-N,N-dimethyltryptamine (4-AcO-DMT) is a synthetic tryptamine derivative with the systematic IUPAC name 3-[2-(dimethylamino)ethyl]-1H-indol-4-yl acetate . Its molecular formula is C₁₄H₁₈N₂O₂, and it has a molecular weight of 246.30 g/mol . The compound features an indole ring substituted at the 4-position with an acetoxy group (-OAc) and a dimethylaminoethyl side chain at the 3-position (Figure 1).
Key Structural Features:
- Indole core: A bicyclic structure comprising a benzene ring fused to a pyrrole ring.
- 4-Acetoxy group: An acetylated hydroxyl group at the 4-position of the indole.
- N,N-Dimethylaminoethyl side chain: A two-carbon chain terminating in a dimethylamine group.
4-AcO-DMT is functionally related to psilocin (4-HO-DMT) and psilocybin (4-PO-DMT), differing primarily in the ester group at the 4-position. While psilocybin contains a phosphate ester, 4-AcO-DMT substitutes this with an acetate ester . The compound is also known by synonyms such as O-acetylpsilocin, psilacetin, and 4-acetoxy-DMT . Its CAS registry number is 92292-84-7 .
Historical Synthesis and Discovery
4-AcO-DMT was first synthesized in 1963 by Albert Hofmann and Franz Troxler during investigations into psilocin derivatives at Sandoz Laboratories . Hofmann’s work aimed to explore structure-activity relationships among tryptamines, building on his earlier isolation of psilocybin from Psilocybe mushrooms. The initial synthesis involved acetylation of psilocin using acetic anhydride, though yields were suboptimal .
In 1999, David E. Nichols and colleagues at Purdue University developed an improved synthetic route to 4-AcO-DMT, enabling higher purity and scalability . This method utilized a Friedel-Crafts acetylation of 4-hydroxyindole followed by N-alkylation with dimethylaminoethyl chloride. Nichols proposed 4-AcO-DMT as a cost-effective alternative to psilocybin for pharmacological studies, citing challenges in synthesizing psilocybin’s phosphate ester .
The compound gained prominence in the 2010s as a research chemical and later as a recreational substance, often marketed as “synthetic psilocybin” due to its prodrug relationship to psilocin .
Significance in Psychedelic Pharmacology Research
Prodrug Mechanism and Metabolic Activation
4-AcO-DMT is rapidly deacetylated in vivo by esterases to yield psilocin (4-HO-DMT), the active metabolite responsible for its psychedelic effects . This bioconversion mirrors the metabolic pathway of psilocybin, which is dephosphorylated to psilocin. Comparative studies in rodents demonstrate that 4-AcO-DMT and psilocybin exhibit equivalent behavioral effects at equimolar doses, confirming their shared mechanism .
Receptor Pharmacology
As a serotonin receptor agonist, 4-AcO-DMT (via psilocin) primarily targets 5-HT₂A receptors, with additional activity at 5-HT₁A, 5-HT₂B, and 5-HT₂C subtypes (Table 1) . Functional assays reveal partial agonism at 5-HT₂A (EC₅₀ = 2.4–3.8 µM) and high efficacy at 5-HT₂C (Eₘₐₓ = 95.1%) . These interactions underpin its hallucinogenic properties and differentiate it from non-psychedelic serotonergics.
Table 1: Receptor Affinity Profile of Psilocin (4-HO-DMT)
| Receptor | Affinity (Kᵢ, nM) | Efficacy (Eₘₐₓ, %) |
|---|---|---|
| 5-HT₂A | 6.0–340 | 16–98 |
| 5-HT₂B | 4.6–410 | 38–84 |
| 5-HT₂C | 10–141 | 95.1 |
| 5-HT₁A | 49–567 | ND |
Data compiled from in vitro studies .
Structural and Crystallographic Insights
X-ray crystallography of 4-AcO-DMT fumarate salts has elucidated its solid-state conformation, revealing planar indole systems and hydrogen-bonding networks critical for receptor interactions . These structural studies inform drug design efforts aimed at optimizing psychedelics for therapeutic applications.
Research Applications
4-AcO-DMT’s stability and ease of synthesis have made it a valuable tool in neuropharmacology. Recent studies utilize it to investigate:
- Neuroplasticity: Mechanisms underlying psychedelic-induced synaptic remodeling .
- Behavioral Models: Head-twitch response (HTR) in rodents as a proxy for human hallucinogenic effects .
- Receptor Dynamics: Cryo-EM structures of 5-HT₂A complexes with psilocin derivatives .
Its legal status as an unregulated analog in some jurisdictions has further facilitated preclinical research, though regulatory landscapes are evolving .
Buy 4-AcO-DMT Online
We provide high purity 4-AcO-DMT (O-Acetylpsilocin) for laboratory and research purposes. Our synthetic tryptamine undergoes rigorous testing to ensure superior quality. Available in powder and fumarate forms, this compound is ideal for studying the effects of prodrug psychedelics on the central nervous system. We ship discreetly to the USA, UK, Germany, and worldwide. Buy 4-AcO-DMT online today to advance your scientific inquiries with guaranteed satisfaction.
What Is 4-AcO-DMT? Definition, CAS Number & Chemical Profile
4-AcO-DMT (Psilacetin, O-Acetylpsilocin) is a synthetic tryptamine and a prodrug of psilocin, the active metabolite of psilocybin (the compound found in “magic mushrooms”). It was first synthesized in 1963 by Albert Hofmann and Franz Troxler and later improved by David E. Nichols in 1999 as a more economical alternative to psilocybin for research purposes.
- CAS Number: 92292-84-7 (freebase), 2748484-99-1 (hydrochloride), 1217230-42-6 (fumarate)
- Chemical Structure: Features an acetoxy group attached to the tryptamine backbone, differentiating it from psilocybin’s phosphate ester.
- Mechanism of Action: Acts as a non-selective serotonin receptor agonist, primarily targeting the 5-HT2A receptor, which mediates its psychedelic effects.
Chemical and Pharmacological Profile of O-Acetylpsilocin
O-Acetylpsilocin belongs to the tryptamine family, characterized by a core indole ring structure. The chemical formula is C14H18N2O2, and it features a CAS number of 92260-49-0. This specific identifier helps researchers verify the exact compound they are analysing.
In terms of pharmacology, 4-AcO-DMT functions primarily as a serotonergic psychedelic. It interacts with the 5-HT2A receptor subtype. This interaction is largely responsible for the psychedelic effects observed in clinical and observational settings. Unlike many other stimulants or depressants, this compound does not typically affect dopamine or norepinephrine systems to the same degree.
We ensure that every batch we supply meets strict analytical standards. Researchers can review the chemical profile on databases like DrugBank to cross-reference structural data. Understanding the pharmacokinetics, such as the onset time and duration of effects, is vital for any legitimate study involving this material.
Chemical Summary Table
| Property | Detail |
|---|---|
| IUPAC Name | 3-(2-acetyloxyethyl)-1H-indol-4-yl dimethylamine |
| CAS Number | 92260-49-0 |
| Chemical Formula | C14H18N2O2 |
| Molar Mass | 246.31 g/mol |
| Form | White powder (HCl or Fumarate) |
| Class | Tryptamine / Psychedelic |
| Solubility | Soluble in water and ethanol |
Mechanism of Action: How Does 4-AcO-DMT Work?
4-AcO-DMT works by acting as a prodrug for psilocin. Once ingested, enzymes in the body remove the acetyl group, converting it into psilocin.
Psilocin then binds to and activates serotonin receptors in the brain, specifically the 5-HT2A receptor. This receptor is crucial for regulating mood, cognition, and perception. By agonizing this receptor, the substance alters the brain’s default mode network, leading to changes in thought patterns and visual processing.
According to information found on Drugs.com, similar compounds are used to study neural pathways. Understanding this mechanism is why many scientists look to buy 4-AcO-DMT online. It provides a consistent and reproducible way to stimulate these receptors without the variability of organic mushrooms.
The specificity of 4-AcO-DMT for the 5-HT2A receptor makes it a key candidate for researching treatments for depression and anxiety disorders, although clinical trials are still ongoing.
How Does 4-AcO-DMT Work?
4-AcO-DMT is rapidly metabolized into psilocin by esterase enzymes in the body, with over 99.9% conversion within 5 minutes in human plasma. This makes it functionally identical to psilocybin in terms of psychoactive effects, but with a faster onset and potentially smoother experience.
Comparison to Psilocybin and DMT
| Feature | 4-AcO-DMT | Psilocybin | DMT |
|---|---|---|---|
| Onset | 20-40 minutes | 30-60 minutes | Seconds (smoked) |
| Duration | 4-8 hours | 4-6 hours | 5-30 minutes |
| Nausea | Reduced | Common | Rare |
| Visuals | Bright, geometric | Earthy, organic | Intense, fractal |
| Legal Status | Gray area | Schedule I (USA) | Schedule I (USA) |
Source: PsychonautWiki, ACS Lab
Dosage Guide: How Much 4-AcO-DMT Should You Take?
Dosage ranges for 4-AcO-DMT vary by experience level and route of administration:
| Experience Level | Oral Dose (mg) | Insufflated Dose (mg) | Effects |
|---|---|---|---|
| Threshold | 2-5 | 1-3 | Mild sensory enhancement |
| Light | 5-10 | 3-8 | Enhanced colors, introspection |
| Common | 10-20 | 8-15 | Full psychedelic experience |
| Strong | 20-30 | 15-25 | Intense visuals, ego loss |
| Heavy | 30+ | 25+ | Overwhelming, mystical |
Note: Insufflation (snorting) results in a faster, more intense onset but shorter duration.
Effects of 4-AcO-DMT:
While our products are for in vitro research, understanding the potential biological effects is necessary for handling them safely. Literature from Tripsitter suggests that physiological effects can include mydriasis (pupil dilation), increased heart rate, and nausea.
Psychological effects may include anxiety, confusion, or ego dissolution if the substance is mishandled. In a laboratory setting, researchers must wear appropriate personal protective equipment (PPE). This includes gloves, goggles, and lab coats to prevent accidental exposure.
Physical and Cognitive Effects
- Visuals: Bright, geometric patterns, enhanced colors, and closed-eye imagery.
- Emotional: Euphoria, introspection, empathy, and ego dissolution.
- Body Load: Generally less nausea than psilocybin mushrooms, with a smoother come-up.
- Duration: Effects peak at 2-3 hours and last 4-8 hours.
User Experiences
“4-AcO-DMT felt like a cleaner, more controlled version of mushrooms. The visuals were brighter, and the body load was almost nonexistent.” — PsychonautWiki
Side Effects and Safety Considerations
Common Side Effects
- Psychological: Anxiety, paranoia, or low mood (especially in high doses or unfavorable settings).
- Physical: Mild nausea, increased heart rate, or temporary cognitive impairment.
Who Should Avoid 4-AcO-DMT?
- Individuals with a history of schizophrenia, severe anxiety, or cardiovascular issues.
- Those on MAOIs, lithium, or tramadol (risk of dangerous interactions).
Safety Tip: Always start with a low dose and use a trip sitter for harm reduction.
Legal Status of 4-AcO-DMT
Legal Status by Country
| Country/Region | Legal Status |
|---|---|
| USA | Gray area: Not explicitly scheduled, but may fall under the Federal Analogue Act as a psilocin analog. |
| UK | Class A (illegal to possess, produce, or supply) under the Misuse of Drugs Act. |
| Germany | Controlled substance (Anlage I) under the NpSG. |
| Australia | Schedule 9 (prohibited substance). |
| Asia | Varies: Generally illegal in most countries (e.g., Japan, China). |
Note: Laws are subject to change. Always check local regulations before purchasing or using 4-AcO-DMT.
Research and Therapeutic Potential
Why Are Researchers Interested in 4-AcO-DMT?
- Cost-Effective Alternative: Easier and cheaper to synthesize than psilocybin, making it ideal for pharmacological research.
- Therapeutic Potential: Early studies suggest it may help with depression, PTSD, and anxiety, similar to psilocybin.
- Reduced Side Effects: Users report less nausea and body load compared to magic mushrooms.
Where to Buy 4-AcO-DMT Online Safely in the UK and USA
Finding a reliable supplier is often the most challenging part of research. At Chem14, we simplify this process. We offer 4-AcO-DMT for sale with a focus on safety, legality, and discretion.
We serve a diverse client base across the United Kingdom, United States of America, Germany, Australia, and Asia. Our platform adheres to strict safety guidelines. We ensure that all transactions are secure and that packaging is discreet to maintain privacy.
When you buy psychedelic research chemicals for sale from us, you receive a Certificate of Analysis. This document verifies the purity of the compound, ensuring that your research results are accurate and reproducible. We advise all customers to review our research ethics and safety guidelines before purchasing.
Legal Status of 4-AcO-DMT in Europe and Australia
The legal landscape for synthetic tryptamines varies significantly by jurisdiction. In the United Kingdom, 4-AcO-DMT is a Class A drug under the Misuse of Drugs Act 1971. This makes it illegal to produce, supply, or import for human consumption.
In the United States, the situation is complex. While not explicitly scheduled at the federal level, it may be considered an analogue of psilocin under the Federal Analogue Act, depending on its intended use. This makes it risky to possess without proper licensing for research.
In Germany and other parts of Europe, it often falls under controlled substance legislation, specifically the NpSG (New Psychoactive Substances Act). Australia classifies it as a Schedule 9 prohibited substance.
We strongly recommend checking local laws before you buy 4-AcO-DMT online. Our products are strictly for laboratory and research use only and are not intended for human consumption. You can read more about the legal status on Wikipedia or through PsychonautWiki.
Potential Side Effects and Safety Considerations
While our products are for in vitro research, understanding the potential biological effects is necessary for handling them safely. Literature from Tripsitter suggests that physiological effects can include mydriasis (pupil dilation), increased heart rate, and nausea.
Psychological effects may include anxiety, confusion, or ego dissolution if the substance is mishandled. In a laboratory setting, researchers must wear appropriate personal protective equipment (PPE). This includes gloves, goggles, and lab coats to prevent accidental exposure.
We prioritize safety at Chem14. Please consult our FAQ section for detailed handling instructions. We advise researchers to keep a neutralising agent nearby and to work in a fume hood when handling powders.
Order 4-AcO-DMT for Sale: Why Choose Chem14?
Choosing the right vendor impacts the quality of your research. We distinguish ourselves through product purity and customer service.
- Verified Purity: We test all batches using GC-MS and HPLC techniques.
- Global Shipping: We deliver to the USA, UK, and worldwide with tracking.
- Secure Payment: We use encrypted payment gateways to protect your data.
- Expert Support: Our team understands chemistry and can answer technical questions.
If you are looking to buy tryptamines for sale, our catalogue includes a wide range of compounds. From 4-AcO-MET to 4-AcO-MIPT, we stock the latest substances needed for modern science.
Research Applications and Uses
The primary application of 4-AcO-DMT is in forensic toxicology and neuroscience. Researchers use it to study receptor binding affinities. It is also used to develop detection methods for novel psychoactive substances in biological samples.
Some academic institutions study its potential as a therapeutic agent. The mechanism involving the 5-HT2A receptor is a target for developing new antidepressants. By studying how this compound alters neural plasticity, scientists hope to understand the biological basis of mood disorders.
We supply to universities, private laboratories, and research institutes. If your institution requires a specific formulation, please contact us. We also offer related products like SGT-78 and 5F-MDA-19 for broader research needs.
Is 4-AcO-DMT the same as psilocybin?
No, but it metabolizes into psilocin, the same active compound as psilocybin. Effects are very similar, though some users report 4-AcO-DMT is smoother and more visual.
Can you microdose 4-AcO-DMT?
Yes. A typical microdose is 1-5 mg, taken every few days. Users report enhanced creativity, focus, and mood without full psychedelic effects.
Where can I buy 4-AcO-DMT online?
You can order 4-AcO-DMT for sale at Chem14.com, a trusted supplier of high-purity research chemicals with worldwide shipping.
Is 4-AcO-DMT legal in the USA?
It exists in a legal gray area. While not explicitly scheduled, it may be considered an analog of psilocin under the Federal Analogue Act.
How long does 4-AcO-DMT last?
Effects typically last 4-8 hours, with the peak occurring at 2-3 hours after ingestion.
Is 4-AcO-DMT the same as magic mushrooms?
They are not the same chemically, but they produce similar effects. 4-AcO-DMT is a synthetic prodrug that converts to psilocin, the same active metabolite found in psilocybin mushrooms.
Where can I buy 4-AcO-DMT online?
You can purchase high quality 4-AcO-DMT directly from Chem14.com. We ensure secure shipping and laboratory grade purity for researchers.
What is the recommended dosage for research?
There is no safe dosage for human consumption. For laboratory analysis, researchers typically handle micrograms to milligrams depending on the assay sensitivity. Always follow safety protocols.
Is 4-AcO-DMT legal in the United Kingdom?
No, in the UK it is classified as a Class A drug. It is illegal to possess or supply it for personal use. However, licensed laboratories may obtain it for specific analytical purposes under strict regulations.
How should I store 4-AcO-DMT?
Store the compound in a cool, dry place away from direct sunlight. Ideally, keep it in a sealed container at -20°C for long term stability to prevent degradation from moisture and heat.
 Key Takeaways
Key Takeaways
- 4-AcO-DMT is a synthetic prodrug of psilocin, offering a smoother, more visual experience than psilocybin mushrooms.
- Dosage ranges from 5-30 mg (oral), with effects lasting 4-8 hours.
- Legal status varies: Check local laws before purchasing.
- Potential therapeutic benefits for mental health, but more research is needed.
- Always prioritize safety: Start low, go slow, and use a trip sitter.
 Ready to Buy 4-AcO-DMT Online?
Ready to Buy 4-AcO-DMT Online?
Explore our premium 4-AcO-DMT for sale at Chem14.com. Fast, discreet shipping to the USA, UK, Germany, Australia, and beyond.

References:
- Wikipedia: 4-AcO-DMT
- PsychonautWiki: 4-AcO-DMT
- Recovered: Risks and Benefits of 4-AcO-DMT
- ACS Lab: 4-AcO-DMT Guide
- Cayman Chem: 4-AcO-DMT (Hydrochloride))
- Recovered: Dosage and Safety
- The Hemp Doctor: Dosage Guide
- Third Wave: Microdosing 4-AcO-DMT
- Psychedelic Science Review: 4-AcO-DMT vs. Psilocybin
- Herb: Is 4-AcO-DMT Legal?
- LegalClarity: 4-AcO-DMT Legal Status
-
4-AcO-DMT Crystals CAS 92292-84-7 – Chemicals Research Lab
4-AcO-DMT Crystals CAS 92292-84-7 for sale online. Buy 4-AcO-DMT or O-Acetylpsilocin, 4-high-temperature-DMT, or Psilacetin: psychedelic tryptamine.
-
Buy 4-Acetoxy-N,N-dimethyltryptamine | 92292-84-7
4-Acetoxy-N,N-dimethyltryptamine, commonly referred to as 4-AcO-DMT or …
Buy Online CAS Number 92292-84-7 – TRC – 4-Acetoxy-N,N …
Purchase online CAS Number 92292-84-7 – TRC – 4-Acetoxy-N,N-dimethyltryptamine. High Quality CRMs, Reference Materials, Proficiency Testing & More at LGC Standards.
-
4-ACETOXY-N,N-DIMETHYLTRYPTAMINE (Cas 92292-84-7) – …
Parchem supplies 4-ACETOXY-N,N-DIMETHYLTRYPTAMINE and a range of specialty chemicals worldwide, CAS# 92292-84-7.
-
Category: Specialty Chemicals
-
-
4-ACETOXY-N,N-DIMETHYLTRYPTAMINE | 92292-84 …
Apr 23, 2023 · 4-ACETOXY-N,N-DIMETHYLTRYPTAMINE (CAS 92292-84-7) …
-
Boiling point: 405.6±35.0 °C (Predicted)
-
solubility: Chloroform (Slightly), Methanol (Slightly)
-
Density: 1.158±0.06 g/cm3 (Predicted)
-
storage temp.: 20°C Freezer, Under inert atmosphere
-
-
buy 4-Aco-DMT | Med Chem
Download Chemical Properties of 4 Aco DMT. Unlike other manufacturers our R&D team really care about its purity is at least 99.99%.
-
4-AcO-DMT Fumarate – Chemagora
SDS available for download here. This product is not approved for human or animal testing in vivo. Please note: Quantities over 1g are considered bulk quantities and may take longer than 2 business …
-
4-AcO-DMT Fumarate, 4-AcO Product – Chemlogix.ca
IUPAC: 3- [2- (Dimethylamino)ethyl]-1H-indol-4-yl acetate. CAS: 92292-84-7. …
-
4-ACO-DMT 50grams – BROMADOL
Sale! The 4-ACO-DMT has been manufactured and tested to meet ISO17025:2005 and Guide 34:2009 guidelines. © Bromadol Research Laboratory.
-
4-Aco-DMT | CAS 92292-84-7 | Chemical-Suppliers
4-Aco-DMT | CAS 92292-84-7 REF CSP465978947913 – structural formula, chemical names, physical and chemical properties, references, safety/hazards/toxicity information, supplier lists, and more.
Reviews
There are no reviews yet.