Buy 5-Methoxytryptamine Cas 608-07-1
Buy 5-Methoxytryptamine Cas 608-07-1
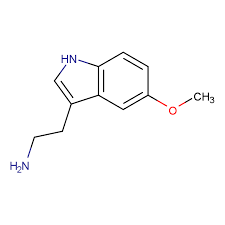
5-Methoxytryptamine (5-MT, 5-MeO-T, or 5-OMe-T), also known as serotonin methyl ether or O-methylserotonin and as mexamine, is a tryptamine derivative closely related to the neurotransmitters serotonin and melatonin.[3] It has been shown to occur naturally in the pineal gland of the brain.[3][4] It is formed via O–methylation of serotonin or N–deacetylation of melatonin.[3][5][4]
5-MT is a highly potent and non-selective serotonin receptor agonist[6][7][8][9] and shows serotonergic psychedelic-like effects in animals.[10] However, it is inactive in humans, at least orally, likely due to rapid metabolism by monoamine oxidase (MAO).[1][2] The levels and effects of 5-MT are dramatically potentiated by monoamine oxidase inhibitors (MAOIs) in animals.[11][12][13][14][15][16]
Use and effects
5-MT is said to produce mild psychoactive effects in humans.[17] It can reportedly potentiate the effects of other drugs such as LSD and THC.[17]
Pharmacology
Pharmacodynamics
| Target | Affinity (Ki, nM) |
|---|---|
| 5-HT1A | 3.2–9 (Ki) 1.1–535 (EC50Tooltip half-maximal effective concentration) 66–135% (EmaxTooltip maximal efficacy) |
| 5-HT1B | 0.75–38 |
| 5-HT1D | 1.7–34 |
| 5-HT1E | 397–3,151 |
| 5-HT1F | 1,166 |
| 5-HT2A | 4.8–724 (Ki) 0.503–0.7 (EC50) 96–119% (Emax) |
| 5-HT2B | 0.51–16 (Ki) 0.7–1.6 (EC50) 99% (Emax) |
| 5-HT2C | 7.1–943 (Ki) 0.1–1.5 (EC50) 100–102% (Emax) |
| 5-HT3 | >10,000 |
| 5-HT4 | 27–2,443 (Ki) 437 (EC50) (pig) 107% (Emax) (pig) |
| 5-HT5A | 45.5 98 (unknown) |
| 5-HT6 | 18–88 |
| 5-HT7 | 0.5–5.0 |
| MT1 | >10,000 |
| MT2 | >10,000 |
| α2A | 1,835 |
| α2B | >10,000 |
| α2C | 2,174 |
| D3 | >10,000 |
| D4 | 1,422 |
| H1, H3 | >10,000 |
| σ1, σ2 | >10,000 |
| KOR | >10,000 |
| SERTTooltip Serotonin transporter | >10,000 4,000 (IC50Tooltip half-maximal inhibitory concentration) 2,169 (EC50) |
| NETTooltip Norepinephrine transporter | >10,000 (IC50) >10,000 (EC50) |
| DATTooltip Dopamine transporter | >10,000 (IC50) 11,031 (EC50) |
| Notes: The smaller the value, the more avidly the drug binds to the site. All proteins are human unless otherwise specified. Refs: [6][7][8][9][18][19][20][21][22] | |
5-MT acts as an agonist of the serotonin 5-HT1, 5-HT2, 5-HT4, 5-HT6, and 5-HT7 receptors.[23][24][25][26][27][28][29][30]
It is an extremely potent serotonin 5-HT2A receptor agonist in vitro, with a EC50Tooltip half-maximal effective concentration of 0.5 nM or 0.7 nM.[8] This was more potent than any other tryptamine evaluated in two large series of compounds.[8][9] For comparison, 5-MeO-DMT had an EC50 of 3.87 nM (7.7-fold lower) and dimethyltryptamine (DMT) had an EC50 of 38.3 nM (76-fold lower).[9]
5-MT has been said to be 25- and 400-fold selective for the serotonin 5-HT2B receptor over the serotonin 5-HT2A and 5-HT2C receptors, respectively.[31] Conversely, one assay reported comparable, low or sub-nanonomolar potencies for all three receptors and the 5-HT1A receptor.[22]
5-MT, in contrast to the closely related melatonin, has no affinity for the melatonin receptors.[32][33] Conversely, in tango assay it displayed agonist-like activity at MT1.[22] However, it may also be converted into melatonin in the body, and hence may indirectly act as a melatonin receptor agonist.[3][5]
5-MT shows dramatically reduced activity as a monoamine releasing agent compared to tryptamine and serotonin.[8]
Effects in animals and humans
5-MT dose-dependently induces the head-twitch response (HTR), a behavioral proxy of psychedelic effects, in rodents, and this effect is reversed by serotonin 5-HT2A receptor antagonists.[10][15][16][34][35][36][37] As such, it may be a hallucinogen in humans.[38] However, in one recent study subcutaneous injection failed to increase HTRs under any conditions in mice, only inducing 5-HT1A-mediated hypothermia and hypolocomotion.[22] 5-MT is only briefly mentioned in several places in Alexander Shulgin‘s TiHKAL and its psychoactive effects are not described.[39][40] Besides psychedelic-like effects, 5-MT produces a “hyperactivity syndrome” in rodents.[3][11][41] It produces various other effects in animals as well.[3]
Pharmacokinetics
Distribution
5-MT is able to cross the blood–brain barrier and enter the central nervous system with peripheral administration in animals.[11] However, it has also been reported that 5-MT shows strong peripheral selectivity in animals comparable to serotonin and bufotenin and that its capacity to exert central effects is limited.[42]
Metabolism
5-MT is metabolized by deamination by monoamine oxidase (MAO), specifically monoamine oxidase A (MAO-A) and to a much lesser extent by monoamine oxidase B (MAO-B).[12][13][14][43]
Brain levels of 5-MT following central administration of 5-MT in rats were potentiated by 20-fold by the MAO-A inhibitor clorgyline and by 5.5-fold by the MAO-B inhibitor selegiline.[13][12] Similarly, levels of serotonin and phenethylamine were also greatly elevated by these drugs.[12][13] In accordance with the potentiation of brain levels of 5-MT by MAOIs, the behavioral effects of centrally administered 5-MT in rats, for instance in the conditioned avoidance response test, are markedly enhanced by MAOIs, including by the dual MAO-A and MAO-B inhibitor iproniazid and by clorgyline and selegiline.[13]
Similarly to rat findings, pineal gland levels of endogenous 5-MT are dramatically elevated by the MAO-A inhibitor clorgyline and by the dual MAO-A and MAO-B inhibitor pargyline in hamsters, and plasma levels of exogenous 5-MT are greatly elevated by these MAOIs as well.[14] Conversely, selegiline was ineffective in elevating brain or plasma 5-MT levels in hamsters.[14]
The non-selective MAO-A and MAO-B inhibitor tranylcypromine has been frequently used to potentiate the effects of 5-MT in animal studies.[11][35][37][15][16]
5-MT is orally inactive in humans presumably due to rapid metabolism by MAO-A.[1][2]
Metabolites of 5-MT include 5-methoxyindole-3-acetic acid (5-MIAA) and 5-methoxytryptophol.[3][14] It may also be metabolized into melatonin.[3][5]
Chemistry
5-MT, also known as 5-methoxytryptamine or as 5-hydroxytrypamine O-methyl ether, is a substituted tryptamine and a derivative of serotonin (5-hydroxytryptamine) and precursor of melatonin (N-acetyl-5-methoxytryptamine).[44]
Properties
The predicted log P of 5-MT is 0.5 to 1.41.[44][45][46]
Analogues and derivatives
5-MT is closely related to other 5-methoxylated tryptamines such as 5-MeO-NMT, 5-MeO-DMT, 5-MeO-DPT, 5-MeO-DiPT, 5-MeO-MiPT, 5-MeO-DALT, and 5-MeO-AMT. 5-MeO-AMT is orally active in humans, in contrast to 5-MT, and could be thought of as a sort of orally active form of 5-MT.[2] Some other notable analogues of 5-MT include tryptamine, 2-methyl-5-hydroxytryptamine, 5-phenoxytryptamine, 5-benzyloxytryptamine, 5-carboxamidotryptamine, 5-methyltryptamine, 5-(nonyloxy)tryptamine, α-methyl-5-hydroxytryptamine, acetryptine (5-acetyltryptamine), and isamide (N-chloroacetyl-5-methoxytryptamine), among others.
Natural occurrence
Biosynthesis
5-MT can be formed by O–methylation of serotonin mediated by hydroxyindole O-methyltransferase (HIOMT) or by N–deacetylation of melatonin.[3][5] It is also a precursor of 5-MeO-DMT in some species.[3]
History
5-MT was encountered online as a reported designer drug by 2023.[17]
Society and culture
Legal status
Canada
5-MT is not a controlled substance in Canada as of 2025.[47]
United States
5-MT is not an explicitly controlled substance in the United States.[48] However, it could be considered a controlled substance under the Federal Analogue Act if intended for human consumption.
Reviews
There are no reviews yet.