Buy Bufotenin Cas 487-93-4
Buy Bufotenin Cas 487-93-4
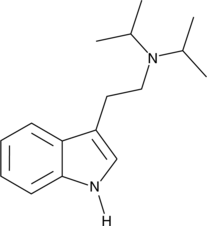
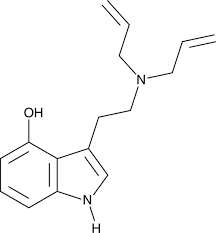
Bufotenin, also known as dimethylserotonin or as 5-hydroxy-N,N-dimethyltryptamine (5-HO-DMT), is a serotonergic psychedelic of the tryptamine family. It is a derivative of the psychedelic dimethyltryptamine (DMT) and of the neurotransmitter serotonin (5-hydroxytryptamine; 5-HT). The compound is an alkaloid found in some species of mushrooms, plants, and toads. It is also found naturally in the human body in small amounts.[7][8][9] Bufotenin, for instance derived from the trees Anadenanthera colubrina and Anadenanthera peregrina, has a long history of entheogenic use as a snuff in South America.[1][5][10][11]
The name bufotenin originates from the toad genus Bufo, which includes several species of psychoactive toads, most notably Incilius alvarius (formerly Bufo alvarius), that secrete bufotoxins from their parotoid glands.[12] However, Bufo and related species like Incilius alvarius contain only trace amounts of bufotenin, with their major active component instead being 5-MeO-DMT. In addition to DMT and serotonin, bufotenin is similar in chemical structure to other psychedelics such as 5-MeO-DMT and psilocin (4-HO-DMT). These compounds also occur in some of the same fungus, plant, and animal species as bufotenin.
Bufotenin acts as a potent and non-selective serotonin receptor agonist, including of the serotonin 5-HT1A, 5-HT2A, 5-HT2C, and 5-HT3 receptors, among others.[13][6][8][14] It also acts as a potent and specific serotonin releasing agent.[14] The compound is more hydrophilic than other related tryptamines and consequently is more peripherally selective.[13][15] In relation to this, bufotenin has been associated with prominent peripheral serotonergic side effects, such as cardiovascular changes.[13][8][16] The cardiovascular effects of bufotenin can be powerful and potentially dangerous.[17]
For many decades and even into the present, bufotenin has been considered by many experts, such as David E. Nichols, to be either inactive or only weakly active as a psychedelic in humans and to produce robust toxic effects.[15][2][13][3] Alexander Shulgin was also uncertain whether bufotenin was an active psychedelic.[16][18][5] However, Jonathan Ott found in 2001 via self-experimentation that bufotenin is in fact a potent psychedelic and does not necessarily produce serious adverse effects.[3][5][2][1] Hamilton Morris has further supported these findings with his own self-experimentation, although bufotenin was reported to be strongly nauseating for himself and many others.[10][5] According to Morris, the psychedelic effects of bufotenin are like a cross between those of DMT and 5-MeO-DMT.[10][5] Morris has stated that bufotenin may in fact be the psychedelic with the longest history of human entheogenic use.[10][5] Bufotenin has also been encountered as a recreational drug in forensic samples, for instance in New York City.[19]
Use and effects
Fabing & Hawkins (1955)
In 1955, Fabing and Hawkins administered bufotenin intravenously at doses of up to 16 mg to prison inmates at Ohio State Penitentiary.[4] A toxic effect causing purpling of the face was seen in these tests.
A subject given 1 mg reported “a tight feeling in the chest” and prickling “as if he had been jabbed by needles.” This was accompanied by a “fleeting sensation of pain in both thighs and a mild nausea.”[4]
Another subject given 2 mg reported “tightness in his throat.” He had tightness in the stomach, tingling in pretibial areas, and developed a purplish hue in the face indicating blood circulation problems. He vomited after 3 minutes.[4]
Another subject given 4 mg complained of “chest oppression” and that “a load is pressing down from above and my body feels heavy.” The subject also reported “numbness of the entire body” and “a pleasant Martini feeling-my body is taking charge of my mind.” The subject reported he saw red spots passing before his eyes and red-purple spots on the floor, and the floor seemed very close to his face. Within 2 minutes these visual effects were gone, and replaced by a yellow haze, as if he were looking through a lens filter.[4]
Fabing and Hawkins commented that bufotenin’s psychedelic effects were “reminiscent of [LSD] and mescaline but develop and disappear more quickly, indicating rapid central action and rapid degradation of the drug”.[4]
Isbell (1956)
In 1956, Harris Isbell at the Public Health Service Hospital in Lexington, Kentucky, experimented with bufotenin as a snuff made from Anadenanthera peregrina.[20] He reported “no subjective or objective effects were observed after spraying with as much as 40 mg bufotenine”; however, subjects who received 10–12 mg by intramuscular injection reported “elements of visual hallucinations consisting of a play of colors, lights, and patterns.”[21][22][20]
Turner & Merlis (1959)
Turner and Merlis (1959)[22] experimented with intravenous administration of bufotenin (as the water-soluble creatinine sulfate salt) to schizophrenics at a New York state hospital. They reported that when one subject received 10 mg during a 50-second interval, “the peripheral nervous system effects were extreme: at 17 seconds, flushing of the face, at 22 seconds, maximal inhalation, followed by maximal hyperventilation for about 2 minutes, during which the patient was unresponsive to stimuli; her face was plum-colored.” Finally, Turner and Merlis reported:
on one occasion, which essentially terminated our study, a patient who received 40 mg intramuscularly, suddenly developed an extremely rapid heart rate; no pulse could be obtained; no blood pressure measured. There seemed to have been an onset of auricular fibrillation . . . extreme cyanosis developed. Massage over the heart was vigorously executed and the pulse returned to normal . . . shortly thereafter the patient, still cyanotic, sat up saying: “Take that away. I don’t like them.”
After pushing doses to the morally admissible limit without producing visuals, Turner and Merlis conservatively concluded: “We must reject bufotenine . . . as capable of producing the acute phase of Cohoba intoxication.”[21]
Hofmann (1963)
Albert Hofmann tried bufotenin orally at doses of up to 50 mg but experienced no psychoactive effects.[23][1]
McLeod and Sitaram (1985)
A 1985 study by McLeod and Sitaram in humans reported that bufotenin administered intranasally at a dose of 1–16 mg had no effect, other than intense local irritation. When given intravenously at low doses (2–4 mg), bufotenin oxalate caused anxiety but no other effects; however, a dose of 8 mg resulted in profound emotional and perceptual changes, involving extreme anxiety, a sense of imminent death, and visual disturbance associated with color reversal and distortion, and intense flushing of the cheeks and forehead.[24]
Shulgin (1997)
Alexander Shulgin reviewed the literature on bufotenin in his book TiHKAL.[16] However, he and his collaborators did not appear to try it themselves.[16]
Ott (2001)
In 2001, ethnobotanist Jonathan Ott published the results of a study in which he self-administered free base bufotenin via insufflation (5–100 mg), sublingually (50 mg), rectally (30 mg), orally (100 mg) and via vaporization (2–8 mg).[2][1] Ott reported “visionary effects” of intranasal bufotenin and that the “visionary threshold dose” by this route was 40 mg, with smaller doses eliciting perceptibly psychoactive effects.[1] He reported that “intranasal bufotenine is throughout quite physically relaxing; in no case was there facial rubescence, nor any discomfort nor disesteeming side effects”.[2][1]
At 100 mg, effects began within 5 minutes, peaked at 35 to 40 minutes, and lasted up to 90 minutes.[2][1] Higher doses produced effects that were described as psychedelic, such as “swirling, colored patterns typical of tryptamines, tending toward the arabesque”.[2][1] Free base bufotenin taken sublingually was found to be identical to intranasal use.[2][1] The potency, duration, and psychedelic action was the same.[2][1] Ott found vaporized free base bufotenin active from 2 to 8 mg with 8 mg producing “ring-like, swirling, colored patterns with eyes closed”.[2][1] He noted that the visual effects of insufflated bufotenin were verified by one colleague, and those of vaporized bufotenin by several volunteers.[2][1]
Ott concluded that free base bufotenin taken intranasally and sublingually produced effects similar to those of Yopo without the toxic peripheral symptoms, such as facial flushing, observed in other studies in which the drug was administered intravenously.[2][1]
Morris (2020s)
Hamilton Morris, a psychoactive drug journalist, the creator of Hamilton’s Pharmacopeia, and a pharmacologist, has experimented with bufotenin and found that it was an active psychedelic.[10][5] He has claimed that its effects are like a cross between those of DMT and 5-MeO-DMT, being less visual than DMT but more visual than 5-MeO-DMT.[10][5] Morris has also stated that bufotenin is very nauseating and this has made it unpleasant for himself and other people.[10][5] By insufflation, he has said that its duration is about 1 hour and is longer than that of DMT or 5-MeO-DMT.[10][5]
Morris and others have suggested use of the serotonin 5-HT3 receptor antagonist ondansetron (Zofran) to prevent nausea and vomiting with especially nauseating or serotonin 5-HT3 receptor agonistic serotonergic psychedelics like bufotenin.[25][26][27]
Side effects
Side effects of bufotenin include nausea and vomiting, among others.[2][1][5][10] It can also produce powerful and potentially dangerous and frightening cardiovascular side effects at doses that allow for hallucinogenic effects.[17]
Overdose
The acute toxicity (LD50) of bufotenin in rodents has been estimated at 200 to 300 mg/kg. Death occurs by respiratory arrest.[28] In April 2017, a South Korean man died after consuming bufotenin-containing toads that had been mistaken for edible Asian bullfrogs,[29] while in Dec. 2019, five Taiwanese men became ill and one man died after eating bufotenin-containing Central Formosa toads that they mistook for frogs.[30]
Reviews
There are no reviews yet.