Buy DMBMPP Cas 1391499-52-7
Buy DMBMPP Cas 1391499-52-7
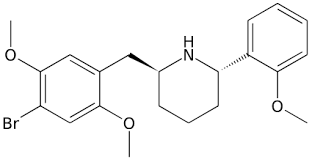
DMBMPP, also known as juncosamine or as 2-(2,5-dimethoxy-4-bromobenzyl)-6-(2-methoxyphenyl)piperidine, is a highly selective serotonin 5-HT2A receptor agonist and 2-benzylpiperidine analogue of the serotonergic psychedelic 25B-NBOMe which is used in scientific research.[1][2][3][4]
Interactions
Pharmacology
Pharmacodynamics
The (S,S)-isomer ((2S,6S)-DMBMPP) is the most selective agonist for the human serotonin 5-HT2A receptor yet discovered, with a affinity (Ki) of 2.5 nM at the human serotonin 5-HT2A receptor and with 124-fold selectivity for the serotonin 5-HT2A receptor over the structurally similar serotonin 5-HT2C receptor.[4][5] Together with 25CN-NBOH,[6] (2S,6S)-DMBMPP is the only known 5-HT2A agonist to exhibit this level of selectivity.[7] In contrast to the case of the serotonin 5-HT2A receptor, no functional data has been reported for DMBMPP at the serotonin 5-HT2C receptor as of 2023.[8][7]
| Ligand | Ki ± SEM (nM) | Ki ± SEM (nM) | Ki ± SEM (nM) |
|---|---|---|---|
| [3H] ketanserin | [3H] mesulergine | fold selectivity | |
| h5-HT2A | h5-HT2C | h5-HT2C/h5-HT2A | |
| 2C-B | 6.0 ± 0.3 | 23.8 ± 2.6 | 9.5 |
| 25B-NBOMe | 0.19 ± 0.01 | 4.0 ± 0.4 | 21 |
| (±)-DMBMPP | 5.3 ± 0.3 | 520 ± 22 | 98 |
| (S,S)-(−)-DMBMPP | 2.5 ± 0.1 | 310 ± 42 | 124 |
| (R,R)-(+)-DMBMPP | 2,100 ± 171 | 28,600 ± 4700 | 27 |
(S,S)-DMBMPP was assessed and found to fully substitute for the psychedelic drug LSD in rodent drug discrimination tests.[9][10][4] As such, DMBMPP may be expected to have hallucinogenic effects in humans.[9][10][4]
Despite its uniquely high selectivity for the serotonin 5-HT2A receptor, it has been said that DMBMPP is not widely used as a pharmacological tool in scientific research, presumably due to its chemical synthesis being relatively inaccessible.[7] Consequently, 25CN-NBOH, another highly selective serotonin 5-HT2A receptor agonist, has been proposed as an alternative to DMBMPP for use in scientific research.[7] DMBMPP and 25CN-NBOH are the two most selective serotonin 5-HT2A receptor agonists known as of 2020.[11]
Chemistry
DMBMPP, also known as 2-(2,5-dimethoxy-4-bromobenzyl)-6-(2-methoxyphenyl)piperidine, is a cyclized phenethylamine, 2C, and NBOMe derivative of 2C-B and 25B-NBOMe.[2] It differs from 25B-NBOMe by incorporating the amine within a piperidine ring, making for a more conformationally restrained, rigid molecular structure than that of the open-chain 25B-NBOMe.[2] The presence of the piperidine ring introduces two stereocenters, thus, four stereoisomers of this compound can be made.[2]
History
DMBMPP was first described in the scientific literature by Jose Juncosa of the lab of David E. Nichols at Purdue University in 2011.[3][4]
Society and culture
Legal status
Canada
DMBMPP is not an explicitly controlled substance in Canada as of 2025.[12] However, it might be covered under phenethylamine and amphetamine blanket-ban language, although this is unclear due to its nature as a cyclized phenethylamine.[12]
United States
DMBMPP is not an explicitly controlled substance in the United States.[13] However, it could be considered a controlled substance under the Federal Analogue Act if intended for human consumption.
Reviews
There are no reviews yet.