Buy N-Methylmescaline (NMM) Cas 4838-96-4
Buy N-Methylmescaline (NMM) Cas 4838-96-4
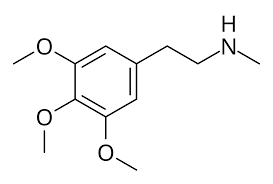
N-Methylmescaline (NMM), also known as methylmescaline (M-M), is an alkaloid and serotonin receptor modulator of the phenethylamine family related to mescaline that occurs naturally in cacti including Lophophora williamsii (peyote), Pelecyphora aselliformis, and Pachycereus pringlei, among others.[1][2][3][4][5]
Use and effects
According to Alexander Shulgin, N-methylmescaline shows no central or peripheral effects at doses of up to 24 or 25 mg, which is many times the minor or trace amounts present in peyote.[6][7][2][8][9][10][11] Nonetheless, according to Shulgin, N-methylmescaline, in combination with potent isoquinoline monoamine oxidase inhibitors (MAOIs) that are also present in the cactus, might be the active psychedelic constituent of Pachycereus pringlei, which notably does not contain mescaline.[3][4][12]
Interactions
Pharmacology
Pharmacodynamics
N-Methylmescaline shows weak affinity for serotonin receptors similarly to mescaline.[1][13] It was found to have about half the serotonin receptor affinity as mescaline (A2 = 5,250 nM vs. 2,240 nM, respectively).[13]
N-Methylmescaline failed to significantly substitute for mescaline (25 mg/kg) in rodent drug discrimination tests either with intraperitoneal or intracerebroventricular injection.[1][14] In contrast to N-methylmescaline however, trichocereine (N,N-dimethylmescaline) produced similar effects to mescaline at a dose of 50 mg/kg intraperitoneally, whereas it only transiently substituted for mescaline when given intracerebroventricularly.[14] Trichocereine has also been reported to be psychedelic in humans, although findings in this area are controversial and conflicting.[7][2][8] It has been noted that N-methylation of psychedelic phenethylamines, for instance Beatrice (N-methyl-DOM), has invariably eliminated their hallucinogenic activity.[15][4]
N-Methylmescaline is less toxic than mescaline in terms of lethal doses in animals.[16]
Chemistry
N-Methylmescaline, also known as N-methyl-3,4,5-trimethoxyphenethylamine, is a substituted phenethylamine and scaline.[1][9] It is specifically the N–methyl analogue of mescaline (3,4,5-trimethoxyphenethylamine).[1][9]
Synthesis
The chemical synthesis of N-methylmescaline has been described.[1]
Analogues
Notable analogues of N-methylmescaline, besides mescaline, include trichocereine (N,N-dimethylmescaline) and N-acetylmescaline.[1][9] Other psychedelic-related N-methylphenethylamines include methyl-TMA (N-methyl-TMA), Beatrice (N-methyl-DOM), N-methyl-DOET, N-methyl-DOB, N-methyl-2C-I, N-methyl-DMA, N-methyl-MMDA-2, and MDMA (N-methyl-MDA).[1][9][15][17]
History
N-Methylmescaline was first described in the scientific literature by Ernst Späth and Johann Bruck in 1937.[4][18] It was isolated from Lophophora williamsii (peyote) by the researchers and was also synthesized.[4]
Society and culture
Legal status
Canada
N-Methylmescaline is not a controlled substance in Canada as of 2025.[19]
United States
N-Methylmescaline is not an explicitly controlled substance in the United States.[1] However, it may be considered controlled in this country as it is a positional isomer of 3,4,5-trimethoxyamphetamine (TMA), which is a specifically regulated substance.[20]
Reviews
There are no reviews yet.