Buy Luvesilocin Cas 2756001-39-3
Buy Luvesilocin Cas 2756001-39-3
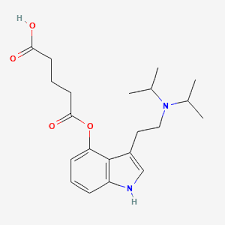
Luvesilocin, also known as RE104 and FT-104, as well as 4-glutaryloxy-N,N-diisopropyltryptamine (4-HO-DiPT O-glutarate or 4-GO-DiPT), is a psychedelic drug of the tryptamine and 4-hydroxytryptamine families which is under development for the treatment of psychiatric disorders.[3][4] It is taken orally or by subcutaneous injection.[3][2]
The drug is a prodrug ester of 4-HO-DiPT, which acts as a non-selective serotonin receptor agonist including of the serotonin 5-HT2A receptor.[5][6]
Luvesilocin was first described in the literature in 2021.[5][7] It is under development for the treatment of postpartum depression and treatment-resistant depression.[8][9][10][11] As of September 2025, the drug has reached phase 2 clinical trials.[12] A phase 3 trial is planned for 2026.[12]
Use and effects

The effects of luvesilocin have been clinically studied.[2] It was evaluated at doses of 5 to 40 mg (equivalent to ~4–32 mg 4-HO-DiPT) by subcutaneous injection in this study.[2] The drug was specifically assessed in terms of modified Drug Effects Questionnaire (DEQ) ratings, Mystical Experience Questionnaire (MEQ) ratings, and adverse effects.[2] The mean duration of the psychedelic experience after administration of luvesilocin at a dose of 30 mg was found to be 3.6 hours.[2][1]
Interactions
Pharmacology
Pharmacodynamics
Luvesilocin is a prodrug that is metabolized into 4-HO-DiPT.[13][14] This metabolite is an analogue of the neurotransmitter serotonin and acts as a non-selective serotonin receptor agonist, including of the serotonin 5-HT2A receptor.[5][6] Activation of the serotonin 5-HT2A receptor is thought to be specifically responsible for the hallucinogenic effects of serotonergic psychedelics.[citation needed]
4-HO-DiPT produces the head-twitch response, a behavioral proxy of psychedelic effects, in rodents.[15] In drug discrimination tests, 4-HO-DiPT fully substituted for the psychedelic drug DOM, with 5-fold lower potency than DOM and 2-fold lower potency than psilocin (4-HO-DMT).[16]
The drug activates basolateral amygdala (BLA) interneurons via the serotonin 5-HT2A receptor to enhance GABAergic inhibition of principal neurons in the BLA, which may mediate an anxiolytic effect of suppression of learned fear (fear extinction) in rodents.[5][6]
Pharmacokinetics
Given by subcutaneous injection, the elimination half-life of luvesilocin is 0.43 to 0.64 hours and of 4-HO-DiPT is 2.7 to 4.1 hours.[2] The mean duration with this route at the employed dose was 3.6 hours.[2]
Chemistry
Synthesis
The chemical synthesis of luvesilocin has been described.[13]
Analogues
Analogues of luvesilocin include 4-HO-DiPT (iprocin), 4-AcO-DiPT (ipracetin), 4-PrO-DiPT, 4-AcO-DMT (psilacetin), 4-PrO-DMT, and 4-GO-DMT (RE-109), among others.
History
Luvesilocin was first described in the literature in 2021.[5][7]
Society and culture
Names
Luvesilocin is the generic name of the drug and its INNTooltip International Nonproprietary Name.[17] It is also known by its developmental code names RE104 or RE-104 and FT104 or FT-104.[3]
Legal status
Canada
Luvesilocin is not a controlled substance in Canada as of 2025.[18]
United States
Luvesilocin is not an explicitly controlled substance in the United States.[19] However, it could be considered a controlled substance under the Federal Analogue Act if intended for human consumption.
Research
Luvesilocin is under development for the treatment of postpartum depression (PPD), treatment-resistant depression, and other psychiatric disorders.[3][1][20][21] As of September 2025, it has reached phase 2 clinical trials for these indications.[12] A phase 3 trial is planned for 2026.[12] The drug is being developed by Reunion Neuroscience (formerly known as Field Trip Health).[3]

Reviews
There are no reviews yet.